
Concepts of Genetics (11th Edition)
11th Edition
ISBN: 9780321948915
Author: William S. Klug, Michael R. Cummings, Charlotte A. Spencer, Michael A. Palladino
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 16PDQ
The study of biochemical mutants in organisms such as Neurospora has demonstrated that some pathways are branched. The data shown in the following table illustrate the branched nature of the pathway resulting in the synthesis of thiamine:
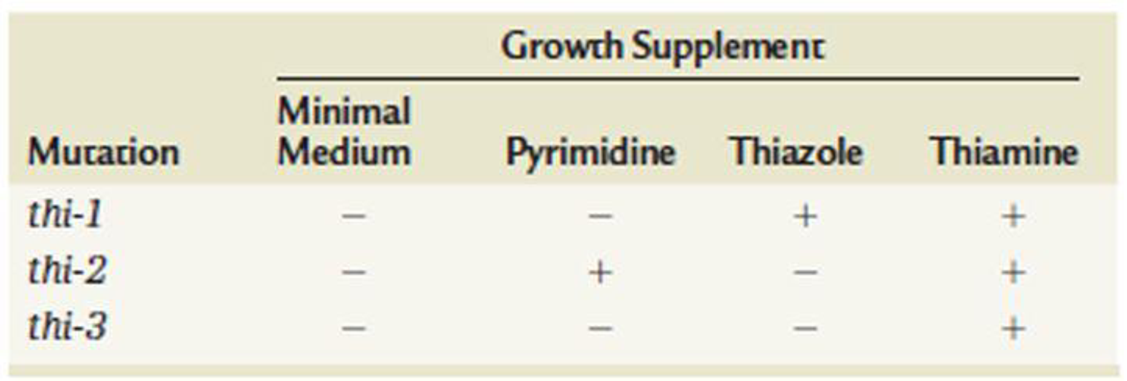
Why don’t the data support a linear pathway? Can you postulate a pathway for the synthesis of thiamine in Neurospora?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Wild-type E. coli cells can synthesize all 20 common amino acids, but some mutants, called amino acid auxotrophs, are unable
to synthesize a specific amino acid and require its addition to the culture medium for optimal growth. Besides their role in
protein synthesis, some amino acids are also precursors for other nitrogenous cell products. Consider the three amino acid
auxotrophs that are unable to synthesize glycine, glutamine, and aspartate, respectively.
Match each nitrogenous product with the mutant or mutants that would fail to synthesize it. Each mutant may fail to synthesize
more than one nitrogenous product, and some nitrogenous products may fail to be synthesized by more than one mutant.
glycine auxotrophs
glutamine auxotrophs
aspartate auxotrophs
Answer Bank
adenine
nucleotides
cytosine
nucleotides
guanine
nucleotides
uridine
nucleotides
An active site of a hypothetical serine protease with a peptide substrate bound is shown below:
This serine protease has 3 specificity pockets (S1, S2, S1') as shown in the figure above. S1 pocket has a glutamic acid in the bottom, the S2 pocket is small and hydrophobic, and the S1' pocket is deep and hydrophobic.
Based on this information you can conclude that R1 is most likely side chain of
a. Asp
b. Lys
c. Lle
d. Gly
e. Phe
Like ribonuclease A, lysozyme from T4 phage is a model enzyme for understanding the energetics and pathways of protein
folding. Unlike ribonuclease A, T4 lysozyme does not contain any disulfide bonds. A number of studies have quantified the
thermodynamic contributions individual amino acid residues and their interactions make to lysozyme folding.
An ion pair between an Asp residue and a His residue in lysozyme contributes 13-21 kJ/mol of favorable folding energy at
pH 6.0. However, this ion pair contributes much less to lysozyme folding at either pH 2.0 or pH 10.0.
Why does the Asp-His ion pair contribute more energy at pH 6.0 than at low or high pH?
At pH 10.0, Asp is protonated and His may be deprotonated.
At pH 6.0, Asp is protonated and His may be deprotonated.
At pH 2.0, Asp and His are both deprotonated.
At pH 10.0, Asp and His are both protonated.
● At pH 6.0, Asp is deprotonated and His may be protonated.
Chapter 14 Solutions
Concepts of Genetics (11th Edition)
Ch. 14 - Prob. 1NSTCh. 14 - A series of mutations in the bacterium Salmonella...Ch. 14 - HbS results from the substitution of valine for...Ch. 14 - Prob. 1CSCh. 14 - Prob. 2CSCh. 14 - Prob. 3CSCh. 14 - HOW DO WE KNOW?
In this chapter, we focused on the...Ch. 14 - CONCEPT QUESTION Review the Chapter Concepts list...Ch. 14 - Contrast the roles of tRNA and mRNA during...Ch. 14 - Francis Crick proposed the adaptor hypothesis for...
Ch. 14 - During translation, what molecule bears the codon?...Ch. 14 - The chain of eukaryotic hemoglobin is composed of...Ch. 14 - Assuming that each nucleotide in an mRNA is 0.34...Ch. 14 - Summarize the steps involved in charging tRNAs...Ch. 14 - To carry out its role, each transfer RNA requires...Ch. 14 - What are isoaccepting tRNAs? Assuming that there...Ch. 14 - Discuss the potential difficulties of designing a...Ch. 14 - Prob. 12PDQCh. 14 - Prob. 13PDQCh. 14 - Prob. 14PDQCh. 14 - The synthesis of flower pigments is known to be...Ch. 14 - The study of biochemical mutants in organisms such...Ch. 14 - Explain why the one-gene: one-enzyme concept is...Ch. 14 - Why is an alteration of electrophoretic mobility...Ch. 14 - Prob. 19PDQCh. 14 - Prob. 20PDQCh. 14 - Prob. 21PDQCh. 14 - Prob. 22PDQCh. 14 - Prob. 23PDQCh. 14 - Several amino acid substitutions in the and ...Ch. 14 - Prob. 25PDQCh. 14 - Prob. 26PDQCh. 14 - Prob. 27PDQCh. 14 - Define and compare the four levels of protein...Ch. 14 - Prob. 29PDQCh. 14 - How does an enzyme function? Why are enzymes...Ch. 14 - Prob. 31PDQCh. 14 - Three independently assorting genes (A, B, and C)...Ch. 14 - How would the results vary in cross (a) of Problem...Ch. 14 - Deep in a previously unexplored South American...Ch. 14 - Prob. 35ESPCh. 14 - The flow of genetic information from DNA to...Ch. 14 - Prob. 38ESPCh. 14 - Knowing the quaternary structure of proteins,...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Many blood clotting proteins undergo a post-translational modification in which specific glutamic acid residues (Glu) in the protein are converted to gamma-carboxyglutamic acid residues (Gla). See reaction scheme below. An example is the blood clotting protein Factor IX, which has 12 Glu in its N-terminus converted to Gla. This modification gives Factor IX the ability to bind calcium and phospholipid membranes. Bacteria do not have the enzyme required to convert Glu to Gla and therefore Factor IX proteins expressed in bacteria would not have the proper modifications. How might you engineer the translational apparatus of a bacterial cell line so that it produces Factor IX with Gla in the appropriate positions. How would you ensure that only the 12 Glu in Factor IX that are normally converted to Gla and not just all Glu (Limit 5-6 senetnces)?arrow_forwardIn a certain bacterial species, the amino acid arginine is synthesized by a particular enzyme so the bacterium does not require arginine in its growth medium. A key enzyme, which we will call arginine synthetase, is necessary for arginine biosynthesis. When these bacteria are given arginine in their growth media, they stop synthesizing arginine intracellularly. Based on this observation alone, propose three different regulatory mechanisms to explain why arginine biosynthesis ceases when arginine is added to the growth medium. To better understand the mechanism of regulation, you measure the amount of intracellular arginine synthetase protein when cells are grown in the presence and absence of arginine. Under both growth conditions, the amount of this protein is identical. Which mechanism of regulation would be consistent with this experimental observation?.arrow_forwardWhich of the following would be a good chemotherapy approach: blocking formationof the ribonucleotide GTP or blocking formation of the deoxyribonucleotide dGTP?Why? Please explain the chemical differences between each of the two nucleotides. Use the specific processes below to support your choice by explaining how either GTP or dGTPare related to these and how loss of the particular molecule would affect each process. *PEP carboxykinase in gluconeogenesis*Succinyl-CoA synthetase in the TCA Cycle*Glucagon signal transductionarrow_forward
- . Mutants of Neurospora crassa that lack carbamoyl phosphate syn- thetase I (CPS I) require arginine in the medium in order to grow, whereas mutants that lack carbamoyl-phosphate synthetase II (CPS II) require a pyrimidine, such as uracil. A priori, one would expect the active CPS II in the arginine mutants to provide sufficient carbamoyl phosphate for arginine synthesis, and the active CPS I in the pyrimidine mutants to "feed" the pyrimidine pathway. Explain these observations.arrow_forwardAs stated in the text, mammalian cells can become resistant to the lethal action of methotrexate by the selective survival of cells con- taining increases in dihydrofolate reductase gene copy number so that intracellular levels of the enzyme become very high. What other biochemical or genetic changes in cells could cause them to become resistant to methotrexate?arrow_forwardThe phylogenetic tree of the OCTN homologs below was generated with the following accession numbers as the basis: NP_957143.1 - OCTN of unknown function CDM63451.1 - OCTN specific for ergothioneine transport XP_021324939.1 - OCTN specific for carnitine transport Which of the Salmo salar homologs has the most probable function as an ergothioneine transporter? Why? asap pleasearrow_forward
- You are working with a protein that is known to form an functioning complex of three identical polypeptide subunits, each with a binding site for a ligand of interest. You are studying an inherited mutation in the protein that changes one Asp residue to a Leu residue in each subunit. A comparison of the binding curves for the normal (wild-type) and mutant versions of the complex are shown below. Suggest a mechanism for the effect that this mutation has on the structure of the complex.arrow_forwardAssume that a series of compounds has been discovered in Neurospora. Compounds A–F appear to be intermediates in a biochemical pathway. Conversion of one intermediate to the next is controlled by enzymes that are encoded by genes. Several mutations in these genes have been identified and Neurospora strains 1–4 each contain a single mutation. Strains 1–4 are grown on minimal media supplemented with one of the compounds A–F. The ability of each strain to grow when supplemented with different compounds is shown in the table (+ = growth; o = no growth). Which biochemical pathway fits the data presented? Media Supplement Strain A B C D E F 1 o o o + + + 2 o o o o + + 3 o o o o + o 4 o o + + + + A) A → B → C → D → E → F B) A → B → C → F → D → E C) F → B → C → D → A → E D) A → B → C → D → F → E E) A → B → F → E → C → Darrow_forwardBacterial species that are capable of synthesising the amino acid histidine do not need it in their growth medium. Histidine biosynthesis requires a specific enzyme, which we shall refer to as histidine synthetase. When these bacteria are supplemented with histidine in their growth media, they cease intracellular histidine synthesis. Propose three distinct regulatory mechanisms to account for why histidine production ends when histidine is present in the growth medium. To further investigate this phenomena, you analyse the quantity of intracellular histidine synthetase protein produced when cells are cultured with and without histidine. The quantity of this protein is same in both scenarios. Which regulatory mechanism makes sense in light of this observation?arrow_forward
- Consider the biochemical pathway shown here. Suppose that a strain of bacteria must synthesize compound 4 to survive and divide. Successful survival and division of bacteria is observed as growth of colonies on an agar plate. This strain of bacteria can grow colonies on minimal medium as long as it is supplemented with compound 1. You are in a lab that has isolated several mutants of this strain. You find that these mutants cannot grow on minimal medium supplemented with compound 1, though they can grow colonies if supplemented with compound 4. Considering what you know about the Beadle-Tatum experiments, which of the following statements would be one that should be true?arrow_forwardChemical modifications of amino acids contribute to the diversity and function of proteins. For instance, γ-carboxylation of specific amino acids is required to make some proteins biologically active. What particular amino acid undergoes this modification, and what is its biological relevance? Warfarin, a derivative of coumarin, which is pres- ent in many plants, inhibits γ-carboxylation of this amino acid and was used in the past as a rat poison. At present, it is also used clinically in humans. What patients might be prescribed warfarin and why?arrow_forwardThe majority of bacterial mutations that need isoleucine also require valine for growth. Why? Which enzyme or process would be deficient in a mutant that requires solely isoleucine for growth (rather than valine)?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
What are Mutations and what are the different types of Mutations?; Author: Science ABC;https://www.youtube.com/watch?v=I16YlE8qTBU;License: Standard youtube license