
Concept explainers
(a)
Interpretation:
The dehydration product of the given alcohol with
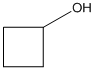
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
(b)
Interpretation:
The dehydration product of the given alcohol with
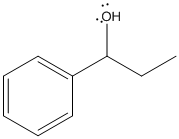
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the alkene formed in major amount is the one which is formed due to the removal of hydrogen from the beta carbon atom with a smaller number of hydrogen atoms. Also, the alkene formed as major product is more substituted.
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
(c)
Interpretation:
The dehydration product of the given alcohol with
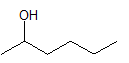
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the alkene formed in major amount is the one which is formed due to the removal of hydrogen from the beta carbon atom with a smaller number of hydrogen atoms. Also, the alkene formed as major product is more substituted.
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
(d)
Interpretation:
The dehydration product of the given alcohol with
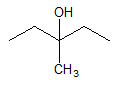
Concept Introduction:
According to Zaitsev's rule, in elimination reaction the alkene formed in major amount is the one which is formed due to the removal of hydrogen from the beta carbon atom with a smaller number of hydrogen atoms. Also, the alkene formed as major product is more substituted.
Dehydration reaction is defined as removal of water molecule from a compound. An alcohol reacts with acid to from alkene as a dehydration product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 1. O-hydroxybenzoic acid is a major product formed with phenol and which other reactant/s I-primary alcohol II-sodium hydroxide III-water IV-carbon dioxide A.I and III B. I and IV C. II and III D. II and IVarrow_forwardWhat products are formed when an alcohol undergoes dehydration?arrow_forwardWhat products are formed when each alcohol is oxidized with K 2Cr 2O 7? In some cases, no reaction occurs.arrow_forward
- Give the structure corresponding to each IUPAC name: a) 3-methyl-3-pentanol b) 4-methyl -2-pentanol c) 2,4 dimethyl -2-hexanol d) 1,3-propanediol e) 3,5-dimethylcyclohexanol f) 6,6-diethyl-4-nonanolarrow_forwardGive the structure corresponding to each IUPAC name. 2,4 dimethyl- 2 hexanolarrow_forwardWhat is the major product formed when each alcohol is treated with HCl?arrow_forward
- Draw the carbonyl products formed when each alcohol is oxidized with K 2Cr 2O 7.arrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions. phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forwardWhat products are formed when each alcohol is oxidized with K2Cr2O7? a. CH3CH2CH2CH2CH2OHarrow_forward
- What alkenes are formed when each alcohol is treated with H 2SO 4? Use the Zaitsev rule to predict the major product.arrow_forwardDescribe chemical properties of alcohols (intermolecular dehydration, intramolecular dehydration).arrow_forwardWhat alcohol is formed when each compound is treated with NaBH4 in CH3OH?arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

