
Concept explainers
(a)
Interpretation:
The
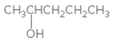
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
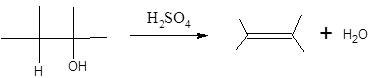
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
(b)
Interpretation:
The alkene formed when the given alcohol is treated with H2SO4 should be determined. The major product should be predicted using Zaitsev rule.
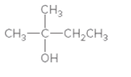
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
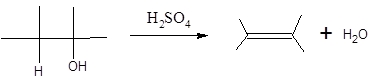
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
(c)
Interpretation:
The alkene formed when the given alcohol is treated with H2SO4 should be determined. The major product should be predicted using Zaitsev rule.
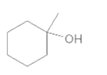
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
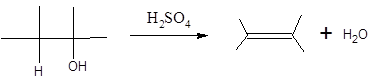
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What products are formed when each alcohol is oxidized with K 2Cr 2O 7? In some cases, no reaction occurs.arrow_forwardDraw the carbonyl products formed when each alcohol is oxidized with K 2Cr 2O 7.arrow_forwardWhat products are formed when benzene is treated with each alkyl chloride and AlCl3?arrow_forward
- What product is formed when the alcohol is oxidized with K2Cr2O7? In some cases, no reaction occurs (if so, draw the given alcohol).arrow_forwardDraw the products formed when each alkene is treated with HCl.arrow_forwardWhat alkenes are formed when each alcohol is dehydrated with TsOH? Label the major product when a mixture resultsarrow_forward
- Draw the organic products formed when each alkyne is treated with two equivalents of HBr.arrow_forwardDraw the structure of a molecule that fi ts each description: a. a 2 ° alcohol of molecular formula C 6H 12O b. a cyclic ether with molecular formula C 5H 10O c. a 1 ° alkyl halide with molecular formula C 5H 11Clarrow_forwardDraw the products formed when ethylene oxide is treated with following reagent. [1] CH3S−; [2] H2Oarrow_forward
