
Concept explainers
Tris(hydroxymethyl)aminomethane, commonly called TRIS or Trizma, is often used as a buffer in biochemical studies. Its buffering range is pH 7 to 9, and Kb is 1.19 × 10−6 for the aqueous reaction
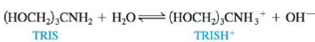
a. What is the optimal pH for TRIS buffers?
b. Calculate the ratio [TRIS]/[TRISH+] at pH = 7.00 and at pH = 9.00.
c. A buffer is prepared by diluting 50.0 g TRIS base and 65.0 g TRIS hydrochloride (written as TRISHCl) to a total volume of 2.0 L. What is the pH of this buffer?
What is the pH after 0.50 mL of 12 M HCl is added to a 200.0-mL portion of the buffer?
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
Chemistry
Organic Chemistry
Organic Chemistry As a Second Language: Second Semester Topics
Principles of Chemistry: A Molecular Approach (3rd Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Two acids, each approximately 0.01 M in concentration, are titrated separately with a strong base. The adds show the following pH values at the equivalence point: HA, pH = 9.5, and HB, pH = 8.5. (a) Which is the stronger acid, HA or HB? (b) Which of the conjugate bases, A or B, is the stronger base?arrow_forwardA buffer is composed of formic acid and its conjugate base, the formate ion. (a) What is the pH of a solution that has a formic acid concentration of 0.050 M and a sodium formate concentration of 0.035 M? (b) What must the ratio of acid to conjugate base be to have a pH value 0.50 units higher than the value calculated in part (a)?arrow_forwardFifty cm3 of 1.000 M nitrous acid is titrated with 0.850 M NaOH. What is the pH of the solution (a) before any NaOH is added? (b) at half-neutralization? (c) at the equivalence point? (d) when 0.10 mL less than the volume of NaOH to reach the equivalence point is added? (e) when 0.10 mL more than the volume of NaOH to reach the equivalence point is added? (f) Use your data to construct a plot similar to that shown in Figure 14.10 (pH versus volume NaOH added).arrow_forward
- A buffer is prepared by mixing 525 mL of 0.50 M formic acid, HCHO2, and 475 mL of 0.50 M sodium formate, NaCHO2. Calculate the pH. What would be the pH of 85 mL of the buffer to which 8.6 mL of 0.15 M hydrochloric acid had been added?arrow_forwardConsider die titration of 50.0 mL of 0.10 M H3A (Ka1 = 5.0 104, Ka2 = 1.0 108, Ka3 = 1.0 1012) titrated by 0.10 M KOH. a. Calculate the pH of the resulting solution at 125 mL of KOH added. b. At what volume of KOH added does pH = 3.30? c. At 75.0 mL of KOH added, is the solution acidic or basic?arrow_forwardAt 25oC, a 1-L buffer solution is prepared by mixing acetic acid and sodium acetate.A) If the solution is to be buffered at pH = 5, what should be the molar ratio of sodium acetate to acetic acid?B) If the buffer is found to be 1.50M in acetic acid and 1.75M in sodium acetate, what will be the pH of the solution?C) If 0.05 mol of NaOH was added to the solution in B, what will be the pH of the solution?D) If the pH of the solution in B is 4.55, how many moles of HCl was added to the solution?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





