
Concept explainers
(a)
Interpretation:
The reactant alcohol should be identified when the
Concept Introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
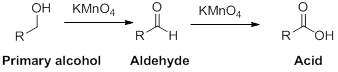
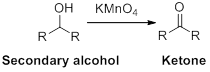
(b)
Interpretation:
The reactant alcohol should be identified when the ketone is the product.
Concept Introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
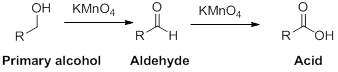
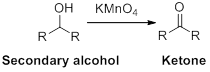
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Fundamentals of General, Organic and Biological Chemistry Volume 1, 5th custom edition for Spokane Community College
- Draw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardHow to separate r and s isomers of tetrahydrofuroic acid by normal phase chromatography?arrow_forwardPropose a scheme to separate and isolate a mixture containing 4-aminobenzoate, 1,2,4,5-tetrachlorobenzene, and napthalene. Isolate two of the three solids by extraction. (please explain what solvents to use and the process step by step)arrow_forward
- The structure for 2-chloro-3-heptene is:arrow_forwardCompare the densities of low-density polyethylene (LDPE) and high-density poly- ethylene (HDPE) with the densities of the liquid alkanes.How might you account for the differences between them?arrow_forwardWhat is the ionisable group of phenazopyridine? With illustrationsarrow_forward
- One of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forwardWhy is this compound achiral?arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
- Which of the alkenes is the most stable?arrow_forwardWith the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forwardPropanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





