
a)
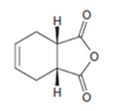
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
b)
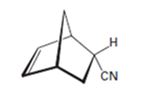
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
c)
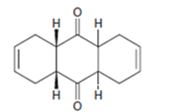
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
d)
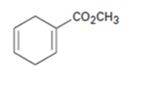
Interpretation:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction are to be stated.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond and if the diene adapts a s-cis conformation during the reaction.
To state:
The diene and dienophile used as starting materials in preparing the compound shown in a Diels-Alder reaction.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Show the product of the Diels–Alder reaction of the following diene with 3-buten-2-one, H2C = CHCOCH3. Make sure you show the full stereochemistry of the reaction product.arrow_forwardWhat diene and dienophile are needed to prepare each compound by a Diels–Alder reaction?arrow_forwardHow many linear dienes have molecular formula C6H10? (Disregard cis–trans isomers.) A. How many of the linear dienes in part a are conjugated dienes? B. How many are isolated dienes? C. How many are cumulated dienes?arrow_forward
- Show the starting diene and dienophile you could use to prepare the following molecule:arrow_forwardThe product can be formed via a Diels Alder reaction. Show the starting materials for the Diels Alder reaction shown below.arrow_forwardShow that the [4 + 2] Diels–Alder reaction is photochemically forbidden.Parrow_forward
- Draw the products of each reaction. Indicate the stereochemistry of Diels–Alder products.arrow_forward21. (a) Predict the product of the following Diels-Alder reaction( Show the correct stereochemistry for the product). cyclopentadiene + cis 1,2-dicyanoethylene (NC-CH=CH-CN) -------> (b) Show the mechanism for the reaction in(a) above.arrow_forwardFor each of the reactions below, the product can be formed via a Diels Alder reaction. Show the starting materials for each of the Diels Alder reactions shown below.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

