
(a)
Interpretation:
The order of the reaction should be determined with respect to oxygen atoms.
Concept Introduction:
Rate Law can be expressed as an integrated rate law and a differential rate law.
Differential Rate Law: This describes the change in the concentrations of reactant as a function of time.
Integrated Rate Law: This describes the initial concentrations and the measured concentration of one or more reactants as a function of time.
(a)
Answer to Problem 36E
The reaction is first order reaction with respect to oxygen.
Explanation of Solution
Given information:
Data is given as:
| Time (s) | |
| 0 | |
The order of reaction can be determined by plotting the graph between
For first order reaction:
For second order reaction:
For zero order reaction:
The given reaction is:
Now, if the reaction is first order with respect to oxygen, then the integrated law is expressed as:
If the reaction is second order with respect to oxygen, then the integrated law is expressed as:
If the reaction is zero order with respect to oxygen, then the integrated law is expressed as:
| Time (s) | ||
| 0 | 22.3327 | |
| 21.36512 | ||
| 20.3376 | ||
| 19.33697 |
The graph between
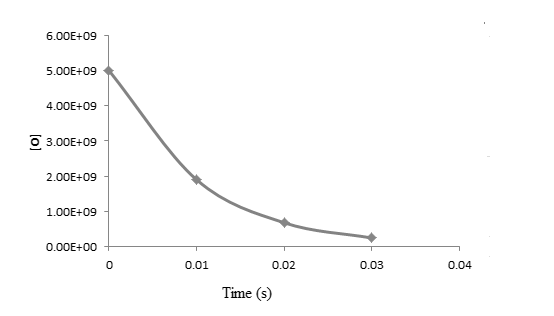 The graph between
The graph between
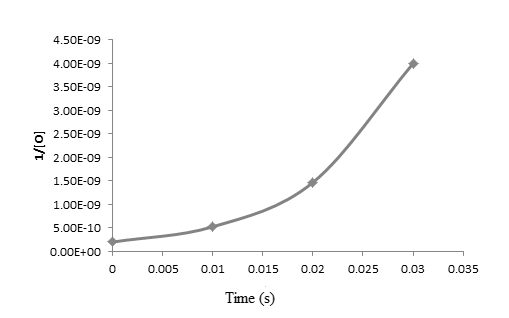
The graph between
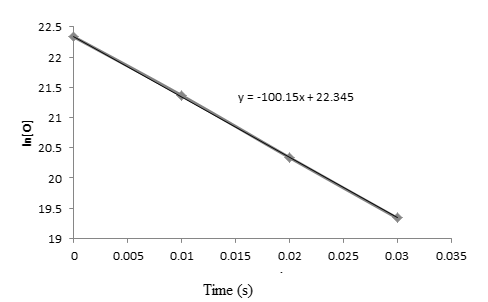
From the above graphs, it is clear that the reaction is first order reaction with respect to oxygen as the graph is straight line graph.
(b)
Interpretation:
The overall rate law and value of rate constant should be calculated.
Concept Introduction:
Rate Law can be expressed as an integrated rate law and a differential rate law.
Differential Rate Law: This describes the change in the concentrations of reactant as a function of time.
Integrated Rate Law: This describes the initial concentrations and the measured concentration of one or more reactants as a function of time.
(b)
Answer to Problem 36E
Rate law is expressed as:
Since, concentration of nitrogen dioxide is more in comparison to oxygen, thus, rate law is written as:
Rate constant for first order reaction is
Explanation of Solution
Given information:
Data is given as:
| Time (s) | |
| 0 | |
The order of reaction can be determined by plotting the graph between
For first order reaction:
For second order reaction:
For zero order reaction:
The given reaction is:
Rate Law for first is expressed as:
Since, it is given that nitrogen dioxide is present in large amount in comparison to oxygen.
Thus, rate law is expressed as:
Where,
Now, from the graph slope is given as:
Slope =
Put the values from graph,
Slope =
Slope =
Thus, value of
Now,
Put the values,
Thus, rate constant for first order reaction is
Want to see more full solutions like this?
Chapter 15 Solutions
Chemical Principles
- The decomposition of gaseous dimethyl ether at ordinary pressures is first-order. Its half-life is 25.0 minutes at 500 C: CH3OCH3(g) CH4(g) + CO(g) + H2(g) (a) Starting with 8.00 g of dimethyl ether, what mass remains (in grams) after 125 minutes and after 145 minutes? (b) Calculate the time in minutes required to decrease 7.60 ng (nanograms) to 2.25 ng. (c) What fraction of the original dimethyl ether remains after 150 minutes?arrow_forwardThe decomposition of iodoethane in the gas phase proceeds according to the following equation: C2H5I(g)C2H4(g)+HI(g) At 660. K, k = 7.2 104 sl; at 720. K, k = 1.7 102 sl. What is the value of the rate constant for this first-order decomposition at 325C? If the initial pressure of iodoethane is 894 torr at 245C, what is the pressure of iodoethane after three half-lives?arrow_forwardIodomethane (CH3I) is a commonly used reagent in organic chemistry. When used properly, this reagent allows chemists to introduce methyl groups in many different useful applications. The chemical does pose a risk as a carcinogen, possibly owing to iodomethanes ability to react with portions of the DNA strand (if they were to come in contact). Consider the following hypothetical initial rates data: [DNA]0 ( mol/L) [CH3I]0 ( mol/L) Initial Rate (mol/Ls) 0.100 0.100 3.20 104 0.100 0.200 6.40 104 0.200 0.200 1.28 103 Which of the following could be a possible mechanism to explain the initial rate data? MechanismIDNA+CH3IDNACH3++IMechanismIICH3ICH3++ISlowDNA+CH3+DNACH3+Fastarrow_forward
- 11.17 Ammonia can react with oxygen to produce nitric oxide and water: 4NH3(g)+5O2(g)4NO(g)+6H2O(g) If the rate at which ammonia is consumed in a laboratory experiment is 4.23 ×10-4 mol L_1s_l, at what rate is oxygen consumed? At what rate is NO produced? At what rate is water vapor produced?arrow_forwardThe half-life of tritium, 3H, is 12.26 years. Tritium is the radioactive isotope of hydrogen. (a) What is the rate constant for the radioactive decay of tritium, in y1 and s1? (b) What percentage of the original tritium is left after 61.3 years?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





