
(a)
Interpretation:
To identify the
Concept introduction:
The synthesis of primary amine is done by using phthalimide and primary alkyl halide in presence of hydroxide base. The important point for this reaction is the phthalimide group has only one hydrogen atom which is attached to nitrogen and can be replaced by alkyl group. Therefore, only one alkyl group can be substitued to the nitrogen atom and so only primary amine will form as the product. The general reaction equation is written as,
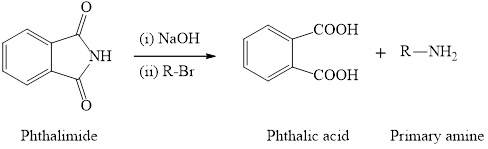
(b)
Interpretation: To identify the alkyl halide used for the preparation of following amines by Gabriel synthesis.
Concept introduction: The synthesis of primary amine is done by using phthalimide and primary alkyl halide in presence of hydroxide base. The important point for this reaction is the phthalimide group has only one hydrogen atom which is attached to nitrogen and can be replaced by alkyl group. Therefore, only one alkyl group can be substitued to the nitrogen atom and so only primary amine will form as the product. The general reaction equation is written as,
(c)
Interpretation:
To identify the alkyl halide used for the preparation of following amines by Gabriel synthesis.
Concept introduction:
The synthesis of primary amine is done by using phthalimide and primary alkyl halide in presence of hydroxide base. The important point for this reaction is the phthalimide group has only one hydrogen atom which is attached to nitrogen and can be replaced by alkyl group. Therefore, only one alkyl group can be substitued to the nitrogen atom and so only primary amine will form as the product. The general reaction equation is written as,
(d)
Interpretation:
To identify the alkyl halide used for the preparation of following amines by Gabriel synthesis.
Concept introduction: The synthesis of primary amine is done by using phthalimide and primary alkyl halide in presence of hydroxide base. The important point for this reaction is the phthalimide group has only one hydrogen atom which is attached to nitrogen and can be replaced by alkyl group. Therefore, only one alkyl group can be substitued to the nitrogen atom and so only primary amine will form as the product. The general reaction equation is written as,
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK ORGANIC CHEMISTRY
- Write the mechanism for the following reactions: 1. the acid-catalyzed hydrolysis of an imine to a carbonyl compound and a primary amine 2. the acid-catalyzed hydrolysis of an enamine to a carbonyl compound and a secondary amine a. How do the two mechanisms differ?arrow_forward1. Why is acetanilide no longer available as a drug in the market? 2. Why is the synthesis of acetanilide still relevant? 3. Can acetanilide be re-offered to market again?arrow_forward1. What are the chemical hazards of both aniline and acetic anhydride? 2. What are the medicinal and industrial uses/application of acetanilide? 3. What was the undesirable effect of acetanilide that made it toxic? 4. How does acetanilide produce an analgesic effect in the body?arrow_forward
- What is the name of the product formed as a result of the reaction between benzylamine and ethylacrylate (ethylpropenoate)? A. N-benzylpropenamideB. N-phenylpropanamideC. N-phenylpropenamideD. Ethyl-3-aminobenzylpropanoateE. N-Ethyl-N-benzylpropanamidearrow_forwardVI. What ammonium salt is formed when each amine is treated with HCI? Draw the structure of the resulting salt. a. -NH2 b. -CH,NHCH3arrow_forwardWhat two amides are obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of propylamine? a. Why is only one amide obtained from the reaction of acetyl chloride with an equivalent of ethylamine and an equivalent of triethylamine?arrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forwardDraw the structure of the amine salt produced when each of the following amines reacts with HCl. 6-50 a. CH3-CH2-CH2-NH2 b. CH3-NH-CH3 CH3 c. CH-CH-CH2-NH2 d. NH-CH-CHarrow_forwardTwo reactions occur when sodium hydroxide is added to methyl salicylate. One is immediate and one only occurs with reflux over time. What type of reaction occurs immediately and with which functional group on methyl salicylate does it react? What type of reaction occurs with reflux over time and with which functional group on methyl salicylate does it react?arrow_forward
- What acyl chloride and amine are required to synthesize the following amides?a. N-ethylbutanamide b. N,N-dimethylbenzamidearrow_forwarda. Write the mechanism for the following reactions:1. the acid-catalyzed hydrolysis of an imine to a carbonyl compound and a primary amine2. the acid-catalyzed hydrolysis of an enamine to a carbonyl compound and a secondary amine b. How do the two mechanisms differ?arrow_forwardIn the 1880's, Acetanilide, sold under the name Antifebrin, was widely used as a pain reliever and fever reducer. However, it had many adverse side effects, including cyanosis as a result of methemoglobinemia. The toxic side effects were the result of a small portion of acetanilide being hydrolyzed to aniline. Acetanilide was discontinued and replaced with phenacetin. Later studies show that both acetanilide and phenacetin are metabolized to acetaminophen. This metabolite, which we know as Tylenol, is responsible for the analgesic and antipyretic properties. Part 1: Show a detailed arrow pushing mechanism of the acid catalyzed hydrolysis of acetanilide to aniline Part 2: Propose a synthesis of Acetaminophen from phenol NH NH NH Phenacetin inophen Acetanilide Attach File Browse Local Files Browse Content Collectionarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


