
(a)
Interpretation: The species which are used for the transesterification of esters other than acids has to be identified.
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
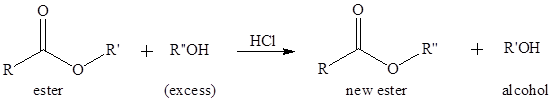
If a better nucleophile is used in place of alcohol molecule that will also increase the rate of transesterification reaction. The conjugate base of an alcohol is an alkoxide which is a better nucleophile and can attack the carbonyl carbon even its oxygen is not protonated.
(b)
Interpretation: The rate of aminolysis reaction of esters does not increase in presence of
Concept introduction: The reaction of a
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK ORGANIC CHEMISTRY
- The reaction of an ester with an amine is not as slow as the reaction of an ester with water or an alcohol. Explain with reason. Explain why the rate of aminolysis of an ester cannot be increased by H+, OH- or OR-. How can you activate the carboxylic acid? Is acid catalyzed hydrolysis of acetamide a reversible or an irreversible reaction. Explain.arrow_forwardThe final products for the hydroxide-ion-promoted hydrolysis of an ester re the carboxylate ion and methanol instead of the carboxylic acid and methoxide ion. Why?arrow_forward1. Amides are comparably unreactive to nucleophilic acyl substitution, yet the B-lactam antibiotics shown) are reactive with a hydroxyl group side chain of an amino acid. Why? R HOarrow_forward
- Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions.b. Explain why it can cross-link DNA.arrow_forward22. The stability of carbocation can be accounted by which of the following structural effect? a. hyperconjugation b. inductive effect d. both a andb C. resonance 23. Secondary amines are more basic than primary amines because of what structural effect? a. hyperconjugation b. inductive effect C. resonance d. steric effectarrow_forwardDefine Nucleophilicity Versus Basicity ?arrow_forward
- Synthesis of p-Bromoaniline Why is the protection of the amine function needed in this reaction? a) The protection increases the selectivity for the ortho substitution. b) The protection increases the reactivity of the reactant in the bromination. c) The protection changes the regioselectivity of reaxtion.arrow_forward5. Make the following via acetoacetic ester synthesis. شد کاarrow_forwardare involved in the regulation of glucose metabolism and in control tissue swelling by the control of inflammation, while regulating cellular salt balance between Na+ and K+. a.glucocorticoids, mineralocorticoids b. mineralocorticoids, glucocorticoids c. androgen, estrogen d. estrogen, androgen Which of the following represents an aldonic acid? OH OH но, но OH но —н но Но HO HO H но но H H- -OH -OH H OH H FOH H- -OH- OH HO но HO. OH II III IV V Refer to the structures. Which of the following compounds will react with acetyl chloride to give an amide and ester, respectively? NH2 ÇOOH CH3 он CHO II II IV V The six groups of enzymes are the following: I. Oxidoreductases II. Transferases III. Hydrolases IV. Isomerases V. Lyases VI. Ligases Fill in the blanks: catalyze hydrolysis while catalyze bond formation.arrow_forward
- Given the information pictured, The final step of the reaction sequence uses PhLi. Provide a reason and possible mechanism by which this final elimination and hydrolysis reaction would occur.arrow_forwardWhy is there a need to add two (2) equivalents of amine in the reaction of an acid chloride and an amine ? A) The extra amine will deprotonate the amine or alcohol, making it a better nucleophile. B) The extra amine will neutralize the acid by-product of the reaction. C) The extra amine will protonate the carbonyl of the acid chloride, making it more reactive. D) The extra amine will absorb the heat of the reaction.arrow_forwardb. Synthesis of imines 1) H+ 2) H2NCH2CH2CH2CH3 3) Н2О ->arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

