
Concept explainers
Interpretation:
To predict the structure of the coloured compound when the given colourless compound come in contact with the acid treated sheet.
Concept introduction:
Acid-base indicators are the compounds which changetheir colour on changing the pH. The reaction of the acid-base indicator can be represented by:
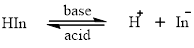
In organic compounds chromophoreis the unsaturated part that imparts colour. An auxochrome is a
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK ORGANIC CHEMISTRY
- propanoic acid + methanol (in concentrated sulfuric acid)arrow_forwardthe fragrant ester benzyl acetate is a naturally occurring compound in many flowers. The flavor and fragrance industry produces this compound by a reaction of benzyl alcohol with excess acetic acid, using hydrochloric acid as a catalyst. Water is a co-product of the reaction. Provide the reaction scheme.arrow_forwardwrite the reactions in which one of the following compounds acts on the ammonia solution of silver oxide. a) propene b) bytine-2 c) cyclopropane d) pentin-1arrow_forward
- Sulphuric acid catalyzes the reaction between compound D and compound E producing propl hexanoate. This reaction is an example of Fischer esterification. Compound D reacts with phosphorus tribromide to give alkyl bromide. Draw the structure and give the name of compound D and Earrow_forwardDefine the term acidic anhydrides?arrow_forwardHydrocarbon A has the molecular formula C14H10C12. It absorbs eight molar equivalents of any hydrogen isotope using a Nickel catalyst. When subjected to ozonolysis, Compound A yields oxalic acid and the chiral chloropentane dioic acid. Write the reactions and propose a structure for A.arrow_forward
- If you attempted to make soap from butanoic acid by forming sodium butanoate, would your preparation have the properties to act as an emulsifying agent (soap)?arrow_forwardPlease answer all parts of the question. Thank you!arrow_forwardWrite the product that will be formed when you obtain Acetyl Chloride (CH3COCl) from Acetic Acid and react with ammonia (NH3arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





