
Concept explainers
a)
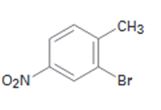
Interpretation:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized is to be shown.
Concept introduction:
The compound required is 2-bromo-4-nitrotoluene. Benzene can be converted into alkyl benzene by Friedal-Crafts alkylation. The alkylbenzene can be nitrated in the ortho and para positions as the alkyl groups are ortho and para directors. Nitro group is deactivating while alkyl group is activating in nature. Hence bromonation of the nitrotoluene will introduce the bromine in the ortho position to alkyl group.
To show:
Starting from benzene and assuming that the ortho and para isomers can be separated how 2-bromo-4-nitrotoluene can be synthesized.
b)
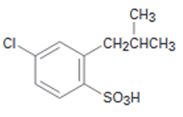
Interpretation:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized is to be shown.
Concept introduction:
The Friedal-Crafts acylation of benzene will yield an acyl benzene as the product. Chlorination of the acyl benzene will produce m-chloroacyl benzene as the acyl group is meta directing. Reduction of the acyl group to the corresponding alkyl group and sulfonation of the product will result in the product required.
To show:
Starting from benzene and assuming that the ortho and para isomers can be separated how the compound can be synthesized.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Show how could you prepare the following compounds starting from benzene as reactant:arrow_forwardGive the skeletal structure of the reacting alkene and the reagents the must eb used to produce BrCH2CHOHCH2Clarrow_forwardHow would you synthesize each of the following compounds from the organic starting material?arrow_forward
- What reagents would you use to prepare each of the following from 3-hexene? a. b.CH3CH2CH2CH2CH2CH3 c. d.arrow_forwardThe addition of water to aldehydes and ketones occurs rapidly, although it is not thermodynamically favored. What would be the product for the reaction above? Hint: Think of the self-ionization of water and the polarity of the carbonyl group.arrow_forwardTo synthesize the following substances using any alkene as a starting material. Name the starting material.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


