
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 19.1, Problem 19.1P
Interpretation Introduction
(a)
Interpretation:
The structural formula of given amide should be drawn.
Concept Introduction:
Amides are organic compounds with −CONH2 as functional group. The general formula of amide is as follows:
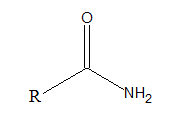
Interpretation Introduction
(b)
Interpretation:
The structural formula of given amides should be drawn.
Concept Introduction:
Amides are organic compounds with −CONH2 as functional group. The general formula of amide is.
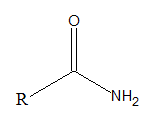
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
PROBLEM 20-1Draw the structures of the following carboxylic acids. trans-2-methylcyclohexanecarboxylic acid
PROBLEM 22-9Show how you would accomplish each conversion using an enamine synthesis withpyrrolidine as the secondary amine.(a) cyclopentanone ¡ 2@allylcyclopentanone(b) pentan@3@one ¡ 2@methyl@1@phenylpentan@3@oneacetophenone Ph C C CH2 Ph
Name the following aldehydes and ketones: (a) 0 (b) A CH2CH2CHO CH3CH2CCHCH3 CH3 (c) 0 0 CH3CCH2CH2CH2CCH2CH3 O (f) (d) CH3 HO АН (e) CH2CH=CHCH2CH2CH H3C. сно HT "CH3 PROBLEM 19-2
Chapter 19 Solutions
Introduction to General, Organic and Biochemistry
Ch. 19.1 - Prob. 19.1PCh. 19.4 - Problem 19-2 Complete the equation for each...Ch. 19.4 - Prob. 19.3PCh. 19 - Prob. 19.4PCh. 19 - Write the IUPAC name for each compound.Ch. 19 - Prob. 19.6PCh. 19 - Prob. 19.7PCh. 19 - Prob. 19.8PCh. 19 - Prob. 19.9PCh. 19 - 0 Complete the equations for these reactions.
Ch. 19 - Prob. 19.11PCh. 19 - Prob. 19.12PCh. 19 - Prob. 19.13PCh. 19 - Prob. 19.14PCh. 19 - Prob. 19.15PCh. 19 - 6 Why are Dacron and Mylar referred to as...Ch. 19 - 7 What type of structural feature do the...Ch. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - 0 Show how triphosphoric acid can form from three...Ch. 19 - 1 Write an equation for the hydrolysis of...Ch. 19 - 2 (Chemical Connections 19A) Locate the ester...Ch. 19 - Prob. 19.23PCh. 19 - Prob. 19.24PCh. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - 8 (Chemical Connections 19C) Once it has been...Ch. 19 - Prob. 19.29PCh. 19 - Prob. 19.30PCh. 19 - Prob. 19.31PCh. 19 - Prob. 19.32PCh. 19 - Prob. 19.33PCh. 19 - 4 (Chemical Connections 19F) Why do Lactomer...Ch. 19 - Prob. 19.35PCh. 19 - Prob. 19.36PCh. 19 - Prob. 19.37PCh. 19 - 8 In Chapter 22, we will discuss a class of...Ch. 19 - Prob. 19.39PCh. 19 - Prob. 19.40PCh. 19 - Prob. 19.41PCh. 19 - Prob. 19.42PCh. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49P
Knowledge Booster
Similar questions
- Problem 16-1 How many hydrogen atoms does pyrrolidine have? How many does purine have? Write the xnolecular formula of each amine.arrow_forwardOhem lab question The experiment is the reduction of a nitro group to an amine I've attached my work, but I'm not sure if it's correct. Can you please double check that my reduction equation is correct and then that my calculations are correct (especially the limiting reactant and theoretical yield) This is the reduction of m-nitroacetophenone and granular tin to m-aminoacetophenone We are using 200 mg of m-nitroacetophenone and 400 mg of granular tin. This will be done with 4mL of 6M hydrochloric acid and 30% sodium hydroxide.arrow_forward18-14 Answer true or false. (a) Carboxylic acids are polar compounds. (b) The most polar bond of a carboxyl group is the C—O single bond. (c) Carboxylic acids have signi?cantly higher boiling points than aldehydes, kebones, and alcohols of comparable molecular weight. (d) The low-molecular-weight carboxylic acids (formic, acetic, propanoic, and butanoic acids) are in?nitely soluble in water. (e) The following compounds are arranged in order of increasing boiling point:arrow_forward
- MULTIPLE CHOICE 1. A 37-40% solution of formaldehyde. * A. butyraldehyde B. acetone C. formalin D. acetaldehyde 2. Which of the following will GIVE a positive reaction to haloform reaction? * A. propanal B. butanal C. pentanal D. acetaldehydearrow_forward18-4 Answer true or false. (a) The functional groups of a carboxylic acid are a carbonyl group bonded to a hydroxyl group. (b) The VSEPR model predicts bond angles of 180° about the carbonyl carbon of a carboxyl group. (c) The VSEPR model predicts bond angles of 109.5° about the oxygen of the OH group of a carboxyl group. (d) The carbonyl carbon of a carboxyl group can be a stereocenter, depending on its location within a molecule. (e) Carboxylic acids can be prepared by chromic acid oxidation of primary alcohols and of aldehydes. (f) The product of chromic acid oxidation of hexanoic acid is 1-hexanol.arrow_forwardWhat structural features of Benadryl are responsible for its biological activity as an H1 antagonist, or anti-histamine? Select all that apply.arrow_forward
- Problem 17-7 Identify all hemiacetals and acetals in the following structures and tell whether each is formed from an aldehyde or a ketone. OH (a) (b) CH3OCH2CH2OCH3 (c)arrow_forwardQuetion 3: which of the following molecules contains an hydroxyl group?arrow_forwardWhat is the correct name for the following compound? Option 1: N-ethyl-N-methyl-1-oxopropanamine Option 2: 1-(ethylamino)-1-(methylamino)propanone Option 3: N-ethyl-N-methylpropanamide Option 4: ethylmethyl propanamide Option 5: 1-(ethylamino)-1-(methylamino)propanalarrow_forward
- Amines Instruction: Propose a synthesis pathway for the following. 1. benzoic acid to benzylamine 2. benzene carboxamide to benzylaminearrow_forwardINSTRUCTION: Select the letter of the correct answer 1. The general reaction between an alcohol and a carboxylic acid is called Alcoholysis Neutralization Dissociation None of the Above In the following fatty acids, the fatty acid with the lowest melting point is Oleic acid Stearic acid Linolenic acid Linoleic acid None of the above The following are non-saponifiable lipids except Carotene Lanolin Cutin Menthol None of the abovearrow_forwardWhat functional group(s) are needed to synthesis the following products (CHOOSE ALL THAT APPLY)Please include the letters with the answers. a. tertiary alcohol b. amide c. primary alcohol d. aldehyde e. secondary alcohol f. carboxylic acid g. aromatic h. ketonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
