
(a)
Interpretation:
Thecomposition of the soap formed by basic hydrolysis of given triacylglycerol needs to be determined.

Concept Introduction:
Lipids are
Triacylglycerol are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
Saponification reaction of triacylglycerol is an alkaline hydrolysis of these triesters that forms sodium or potassium salts of fatty acids (soap) and glycerol as the product. The hydrolysis of triacylglycerol can be done in the presence of aqueous solution of base like
(b)
Interpretation:
The composition of the soap formed by basic hydrolysis of given triacylglycerol needs to be determined.
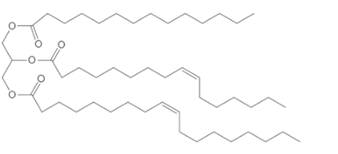
Concept Introduction:
Lipids are biomolecules which are involved in different biochemical reactions.They are special types of organic molecules which can only identify with the help of their physical properties, not by the presence of any certain functional group.
Triacylglycerol are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
Saponification reaction of triacylglycerol is an alkaline hydrolysis of these triesters that forms sodium or potassium salts of fatty acids (soap) and glycerol as the product. The hydrolysis of triacylglycerol can be done in the presence of aqueous solution of base like
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Performing a saponification on a triacylglycerol generates soap. A. True B. False C. Depends on the source of triacylglycerolarrow_forwardThe function of glycerol in embalming fluids is A. to act as a humectant B. to dehydrate C. to preserve D. to preserve body fatsarrow_forwardf) why is significant serum (in the bloodstream) cholesterol reduction so difficult, and why do most of the drugs designed to reduce cholesterol target the liver?arrow_forward
- Draw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B, and C in order of increasing melting point.arrow_forwardDraw the products formed when triacylglycerol A is treated with each reagent, forming compounds B and C. Rank A, B, and C in order of increasing melting pointarrow_forwardWhat is the composition of the soap prepared by hydrolysis of the attachedtriacylglycerol?arrow_forward
- Why is acetone present in pathological urine? What does the presence of acetone indicate?arrow_forwardTo make a soothing chest rub, blend 1 drop each of copaiba oleoresin, black pepper and coriander to _______ of a carrier oil or lotion. Choose one answer. a. 3 tsp b. 2 Tbsp c. 1-2 tsp d. 0.5 tsparrow_forward(a) What are the major components of tea extract? (b) Draw the structures of caffeine, flavonoid, and tannin present in tea.arrow_forward
- Show the saponification of a solid soap.arrow_forwardTrue or False a. Stearic acid is an unsaturated fatty acid.b. Human fat has more unsaturated fatty acids than plant fats.c. Soaps and detergents form micelles when added to water.d. Soap micelles form in water by aggregating the negatively charged carboxylategroups toward the inside and the lipophilic carbon chains toward the outside of themicelle.e. The forces that drive micelle and lipid bilayer formation are the same.f. There are several stereoisomers of cholesterol found in living systems.g. Aldosterone is an androgen.h. Vitamin D precursors have the cholesterol ring system.i. Vitamin E is required for blood clotting.j. Prostaglandins are involved with the inflammatory response.arrow_forwardNormal components of urine Reagents Positive Reaction Uric Acid Indicanarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning



