
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.9B, Problem 1.10P
Use resonance structures to identify the areas of high and low electron density in the following compounds:
- a. H2C=CH—NO2
- b. H2C=CH—C≡N
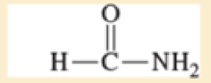
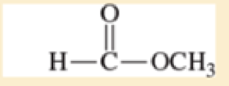
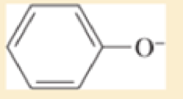
- c. CH3O—CH=CH—CN
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Which of the following is most likely not a gas at room temperature?A) NO2 B) KCl C) CH4 D) CO 2 E) H2
Fluoromethane (CH3F, u=1.81 D) has a smaller dipole moment than chloromethane (CH3Cl u=1.87 D) even though fluorine is more electronegative than chlorine. Explain.
Phosgene, Cl2C = O, has a lower dipole moment than formaldehyde H2C = O. Explain why.
Chapter 1 Solutions
Organic Chemistry (9th Edition)
Ch. 1.2C - a. Nitrogen has relatively stable isotopes...Ch. 1.4 - Draw Lewis structures for the following compounds....Ch. 1.5 - Write Lewis structures for the following molecular...Ch. 1.5 - Circle any lone pairs (pairs of nonbonding...Ch. 1.6 - Use electronegativities to predict the direction...Ch. 1.8 - Prob. 1.6PCh. 1.9B - Draw the important resonance forms for the...Ch. 1.9B - Prob. 1.8PCh. 1.9B - Prob. 1.9PCh. 1.9B - Use resonance structures to identify the areas of...
Ch. 1.10A - Draw complete Lewis structures for the following...Ch. 1.10B - Give Lewis structures corresponding to the...Ch. 1.10B - Prob. 1.13PCh. 1.11 - Compute the empirical and molecular formulas for...Ch. 1.16 - a. Use your molecular models to make ethane, and...Ch. 1.17 - a. Predict the hybridization of the oxygen atom in...Ch. 1.17 - Predict the hybridization geometry and bond angles...Ch. 1.17 - Predict the hybridization, geometry, and bond...Ch. 1.17 - Prob. 1.19PCh. 1.17 - Allene, CH2=C=CH2, has the structure shown below...Ch. 1.17 - 1. Draw the important resonance forms for each...Ch. 1.18B - Prob. 1.22PCh. 1.18B - Two compounds with the formula CH3CH=NCH3 are...Ch. 1.19B - Prob. 1.24PCh. 1.19B - Give the relationship between the following pairs...Ch. 1 - a. Draw the resonance forms for SO2 (bonded OSO)....Ch. 1 - Name the element that corresponds to each...Ch. 1 - Prob. 1.28SPCh. 1 - For each compound, state whether its bonding is...Ch. 1 - a. Both PCl3 and PCl5 are stable compounds Draw...Ch. 1 - Draw a Lewis structure for each species a. N2H4 b....Ch. 1 - Prob. 1.32SPCh. 1 - Prob. 1.33SPCh. 1 - Draw Lewis structures for a. two compounds of...Ch. 1 - Prob. 1.35SPCh. 1 - Some of the following molecular formulas...Ch. 1 - Prob. 1.37SPCh. 1 - Give the molecular formula of each compound shown...Ch. 1 - 1. From what you remember of electronegativities,...Ch. 1 - For each of the following structures, 1. Draw a...Ch. 1 - Prob. 1.41SPCh. 1 - Prob. 1.42SPCh. 1 - Prob. 1.43SPCh. 1 - Prob. 1.44SPCh. 1 - For each pair of ions, determine which on is more...Ch. 1 - Use resonance structures to identify the areas of...Ch. 1 - Prob. 1.47SPCh. 1 - In 1934, Edward A. Doisy of Washington University...Ch. 1 - If the carbon atom in CH2Cl2 were fat. there would...Ch. 1 - Cyclopropane (C3H6, a three-membered ring) is more...Ch. 1 - Prob. 1.51SPCh. 1 - Prob. 1.52SPCh. 1 - In most amines, the nitrogen atom is sp3...Ch. 1 - Predict the hybridization and geometry of the...Ch. 1 - Draw orbital pictures of the pi bonding in the...Ch. 1 - Prob. 1.56SPCh. 1 - Prob. 1.57SPCh. 1 - Which of the following compounds show cis-trans...Ch. 1 - Give the relationships between the following pairs...Ch. 1 - Dimethyl sulfoxide (DMSO) has been used as an...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which molecule in each pair has the greater dipole moment? Give the reason for your choice. (a) ClO2 or SO2 (b) HBr or HCl (c) BeCl2 or SCl2 (d) AsF3 or AsF5arrow_forwardIdentify the orbitals on each of the atoms that form the bonds in H3CCN. How many bonds and bonds form?arrow_forwardKetene, C2H2O, is a reactant for synthesizing cellulose acetate, which is used to make films, fibers, and fashionable clothing. (a) Write the Lewis structure of ketene. Ketene does not contain an OH bond. (b) Identify the electron-region geometry and the molecular geometry around each carbon atom and all the bond angles in the molecule. (c) Identify the hybridization of each carbon and oxygen atom. (d) Is the molecule polar or nonpolar? Use appropriate data to support your answer.arrow_forward
- Consider the following molecules: (a) CH4 (b) NH2Cl (c) BF3 (d) CS2 (i) In which compound are the bonds most polar? (ii) Which compounds are not polar? (iii) Are the H atoms in NH2Cl negative or positive?arrow_forwardMenthol is used in soaps, perfumes, and foods. It is present in the common herb mint, and it can be prepared from turpentine. (a) What are the hybridizations used by the C atoms in the molecule? (b) What is the approximate COH bond angle? (c) Is the molecule polar or nonpolar? (d) Is the six-member carbon ring planar or non-planar? Explain why or why not.arrow_forward(a) Draw a Lewis structure for the ozone molecule, O3. (The order of atom attachment is OOO, and they do not form a ring.) Chemists use ozone to cleave carbon-carbon double bonds (Section 6.5C). (b) Draw four contributing resonance structures; include formal charges. (c) How does the resonance model account for the fact that the length of each OO bond in ozone (128 pm) is shorter than the OO single bond in hydrogen peroxide (HOOH, 147 pm) but longer than the OO double bond in the oxygen molecule (123 pm)?arrow_forward
- What are the bond angles predicted by the VSEPR model about the carbon atom in the formate ion, HCO2? Considering that the bonds to this atom are not identical, would you expect the experimental values to agree precisely with the VSEPR values? How might they differ?arrow_forwardBiotin is being studied in a lab. a. What is the IMF(s) of biotin (C10H16N2O3S)? b. What are the sigma/pi bondings and bond angles of biotin? c. What are the resonance functional groups?arrow_forward7 The pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY