
a)
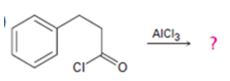
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
b)
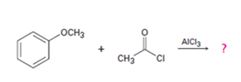
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to be provided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the aromatic ring attacks the elctrophile to give a resonance stabilized intermediate. In the last step the intermediate deprotonates to yield the product. The methoxyl group is an ortho and para orienting group.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
c)
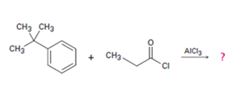
Interpretation:
The products formed in the reaction given are to be identified. The mechanism of the reaction also is to berovided.
Concept introduction:
The mechanism of the electrophilic substitution reaction is to be given. In the first step the electrophile is produced. In the second step the electrons of the aromatic ring attacks the elctrophile to give a resonance stabilized intermediate. In the last step the intermediate deprotonates to yield the product.
To identify:
The products formed in the reaction given and to provide the mechanism of the reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry
