
a)
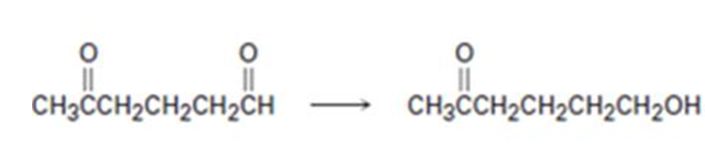
Interpretation:
How to carry out the selective transformation of a ketoaldehyde to ketoalcohol is to be shown.
Concept introduction:
Both
To show:
How to carry out the selective transformation of a ketoaldehyde to ketoalcohol.
b)
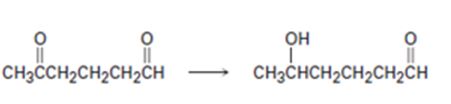
Interpretation:
How to carry out the selective transformation of a diketone to an aldol is to be shown.
Concept introduction:
Both aldehyde and keto groups are reduced by LiAlH4. In order to selectively reduce one keto group, the other keto group is first protected by converting it into an acetal. The protected
To show:
How to carry out the selective transformation of a diketone to an aldol.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- Propose a synthetic route to carry out the following Transformations.arrow_forwardPropose carbonyl group protecting mechanism for the following product *aldehydes and ketones reactionsarrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


