
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.40SP
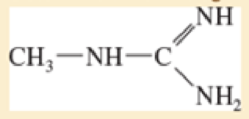
The following compound can become protonated on any of the three nitrogen atoms One of these nitrogens is much more basic than the others, however
- a. Draw the important resonance forms of the products of protonation on each of the three nitrogen atoms
- b. Determine which nitrogen atom is the most basic.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the correct ranking of the structures from highest acidity to lowest acidity for the bolded hydrogen. The correct answer is D, please explain why that is correct and why other options are wrong.
For the following groups of molecules (labeled A-C), rank the acidity of the molecules in order from least acidic (3) to most acidic (1). Explain your reasoning.
Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE.
Arrange the intermediates below in order of increasing basicity:
Chapter 2 Solutions
Organic Chemistry (9th Edition)
Ch. 2.1A - Prob. 2.1PCh. 2.1B - The NF bond is more polar than the NH bond: but...Ch. 2.1B - For each of the following compounds 1. Draw the...Ch. 2.1B - Two isomers of 1,2-dichloroethene are known One...Ch. 2.2C - Prob. 2.5PCh. 2.2C - Prob. 2.6PCh. 2.3 - Prob. 2.7PCh. 2.4 - Calculate the pH of the following solutions a....Ch. 2.6A - Ammonia appears in Table 2-2 as both an acid and a...Ch. 2.7 - Write equations for the following acid-base...
Ch. 2.7 - Ethanol, methylamine. and acetic acid are all...Ch. 2.8 - Prob. 2.12PCh. 2.10 - Write equations for the following acid-base...Ch. 2.10 - Rank the following acids in decreasing order of...Ch. 2.11 - Prob. 2.15PCh. 2.11 - Prob. 2.16PCh. 2.11 - Consider each pair of bases and explain which one...Ch. 2.12 - Which is a stronger base ethoxide ion or acetate...Ch. 2.12 - Prob. 2.19PCh. 2.12 - Prob. 2.20PCh. 2.12 - Prob. 2.21PCh. 2.12 - Choose the more basic member of each pair of...Ch. 2.14 - Prob. 2.23PCh. 2.15D - Classify the following hydrocarbons and draw a...Ch. 2.16D - Prob. 2.25PCh. 2.17C - Draw a Lewis structure and classify each of the...Ch. 2.17C - Circle the functional groups in the following...Ch. 2 - The CN triple bond in acetonitrile has a dipole...Ch. 2 - Prob. 2.29SPCh. 2 - Sulfur dioxide has a dipole moment of 1.60 D....Ch. 2 - Which of the following pure compounds can form...Ch. 2 - Predict which member of each pair is more soluble...Ch. 2 - Prob. 2.33SPCh. 2 - Prob. 2.34SPCh. 2 - Predict which compound in each pair has the higher...Ch. 2 - All of the following compounds can react as acids...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - Rank the following species in order of increasing...Ch. 2 - The Ka of phenylacetic acid is 5 2 105, and the...Ch. 2 - The following compound can become protonated on...Ch. 2 - The following compounds are listed in increasing...Ch. 2 - Prob. 2.42SPCh. 2 - Prob. 2.43SPCh. 2 - Compare the relative acidity of 1-molar aqueous...Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - The following compounds can all react as bases. a....Ch. 2 - The following compounds can all react as acids. a....Ch. 2 - Prob. 2.48SPCh. 2 - Methyllithium (CH3Li) is often used as a base in...Ch. 2 - Label the reactants in these acid-base reactions...Ch. 2 - In each reaction, label the reactants as Lewis...Ch. 2 - Prob. 2.52SPCh. 2 - Each of these compounds can react as a nucleophile...Ch. 2 - Prob. 2.54SPCh. 2 - Give a definition and an example for each class of...Ch. 2 - Circle the functional groups in the following...Ch. 2 - Prob. 2.57SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete each Lewis structure, draw all important resonance structures, predict a value for thebond angles requested, and explain your reasoning. a. Nitrous acid (HNO2)HONOHON=ONO= b. Enolate ion (C2H3O) HC1C2=HC2C1=arrow_forwardA) Indicate the most acidic proton on each molecule. B) Rank in order from least to most which are most acidic C) Draw the enolate formed from the loss of the most acidic proton on the most acidic molecule, Only.arrow_forwardLabel the most acidic hydrogen in each molecule and justify your choice by using appropriate pKa valuesarrow_forward
- The blue N is a nitrogen atom. The numbers are for easy reference to the N atoms. No hydrogens have been drawn in. Is the molecule below aromatic? Explain your answer Which nitrogen atom, 1 or 2, is the more basic N (if you think they are equally basic, write ‘equally basic’)? Explainarrow_forwardIdentify the stronger and weaker Bronsted acid, assign the smaller and largerpKa, identify the stronger and weaker Bronsted conjugate base give an explanation thatincludes resonance contributors if relevant.arrow_forwardWhich is the most acidic proton? Also write down the enolate structure and its resonant structures.arrow_forward
- For the following molecules: circle the most acidic hydrogen(s). determine the approximate pKa of the hydrogen you circled rank the compounds from least (1) to most (5) acidic compound.arrow_forwardFor the following pairs of alcohols, identify the one that is more acidic and explain your choice using ARIO. I said that the molecule on the left side was more acidic due to the e- withdrawing effect, however, my professor stated that resonance takes priority thus the molecule on the right side is the one that is more acidic. Can you explain to me why using ARIO? Thanks in advance!arrow_forward2) For the following groups of molecules (labeled A-C), rank the basicity of the molecules in order from least basic (3) to most basic (1). Explain your reasoning.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY