
(a)
Interpretation:
To draw the complete detailed mechanism and predict the major product
Concept introduction:
Reaction between acid chloride and alcohol to produce ester is named as alcoholysis reaction. In this reaction, addition of alcohol results in breaking of C-Cl bond.
Answer to Problem 21.1P
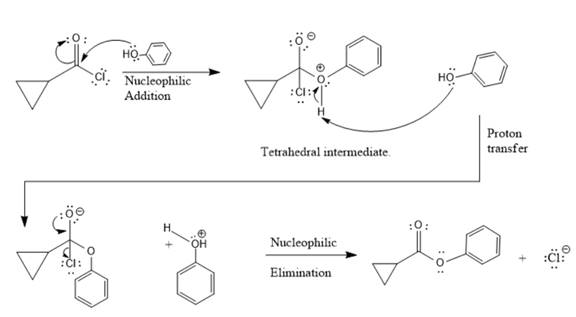
Explanation of Solution
This is an alcoholysis reaction. First step includes nucleophilic addition of alcohol to electrophilic carbon of acid chloride to produce tetrahedral intermediate. This intermediate contains an acidic proton which is rapidly deprotonated by another alcohol molecule. This is via proton transfer. Last step is nucleophilic elimination in which the C=O bond is formed by loss of Cl- group from the intermediate giving an ester.
This is an alcoholysis reaction between an acid chloride and alcohol to give an ester.
(b)
Interpretation:
To draw the complete detailed mechanism and predict the major product
Concept introduction:
Reaction between acid chloride and water to produce carboxylic acid is named as hydrolysis reaction. In this reaction, addition of water molecule results in breaking of C-Cl bond.
Answer to Problem 21.1P
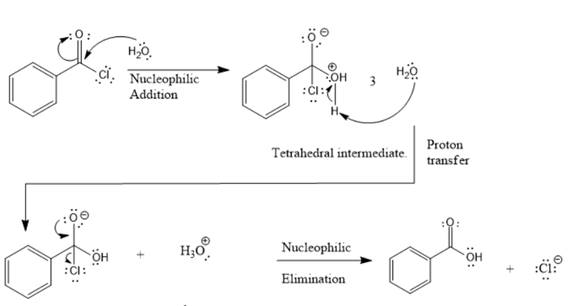
Explanation of Solution
This is a hydrolysis reaction. First step includes nucleophilic addition of water to electrophilic carbon of acid chloride to produce a tetrahedral intermediate. This intermediate contains an acidic proton which is rapidly deprotonated by another water molecule. This is via proton transfer. Last step is nucleophilic elimination in which the C=O bond is formed back by the loss of Cl- group from the intermediate giving carboxylic acid.
This is a hydrolysis reaction between an acid chloride and water to give carboxylic acid.
Want to see more full solutions like this?
Chapter 21 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





