
Concept explainers
Interpretation:
The lock-and-key model for enzymes must be described. The reason for the shapes of the enzyme and its substrate to be important must also be explained.
Concept Introduction:
Enzymes are biocatalysts which perform several biochemical reactions in the cell. It binds selectively with its substrate only though thousands of other molecules are present.
Answer to Problem 26A
Lock-and-key model is a model proposed to explain the selectivity of an enzyme to its substrate.
Shapes of the enzyme and its substrate is important as structural complementarity between the enzyme and its substrate is essential for the binding of the substrate in the active site of the enzyme.
Explanation of Solution
A pictorial representation is shown below to explain lock-and-key model of an enzyme.
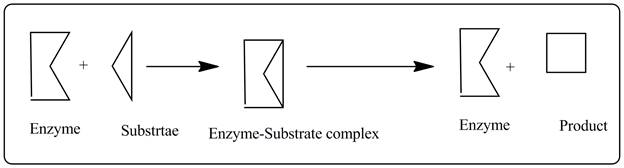
As shown in the picture an enzyme is reacting with a substrate which nicely fits in the active site of the enzyme due to structural complementarity.
This is analogues to a “lock-and-key” as one lock can be unlocked by only one key which is exactly complementary to the lock.
This structural complementarity is responsible for the specificity of an enzyme. The overall shape of the enzyme might be different but the active site of the enzyme must be complementary to the substrate so the substrate can perfectly fits in and biochemical reaction takes place.
Chapter 21 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





