
Concept explainers
Interpretation: Ethers and alcohols needs to be explained as structural isomers using exampled. The functional group in the given table needs to be determined which is the structural isomer of carboxylic acids.
Concept introduction: When two or more molecules have the same formula but shows different properties, they are termed as isomers. Molecules having same molecular formula but arrangement of atoms is different in space are termed as structural isomers.
Answer to Problem 36E
Ethers and alcohols are the structural isomers of one another which can be illustrated by considering a molecular formula
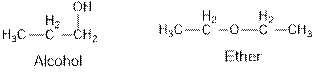
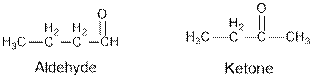
From the given table, esters are the structural isomers of carboxylic acids.
Explanation of Solution
Given information:
| Class | Functional Group | General Formula | Example |
| Halohaydrocarbon | −X (F, Cl, Br, I) | R−X | |
| Alcohols | −OH | R−OH | |
| Ethers | −O− | R−O−R | |
| Aldehydes | 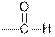 | 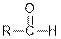 | |
| Ketones | 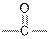 | 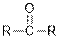 | |
| Carboxylic acids | 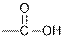 | 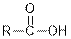 | |
| Esters | 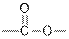 | 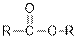 | |
| Amines |
Molecules having same molecular formula but different arrangement of atoms in space are termed as structural isomers.
Ethers and alcohols are the structural isomers of each other which can be illustrated by considering a molecular formula
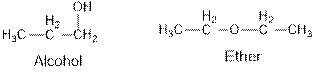
Aldehydes and ketones are the structural isomers of one another which can be illustrated by considering a molecular formula
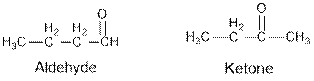
From the given table, esters are the structural isomers of carboxylic acids which can be illustrated by considering a molecular formula
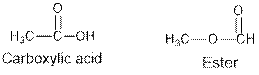
Ethers and alcohols are the structural isomers of each other which can be illustrated by considering a molecular formula
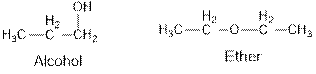
Aldehydes and ketones are the structural isomers of one another which can be illustrated by considering a molecular formula
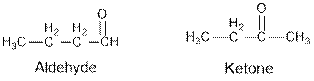
From the given table, esters are the structural isomers of carboxylic acids.
Want to see more full solutions like this?
Chapter 21 Solutions
CHEM.PRINC.W/OWL2+REBATE+2 SUPPL.>IP<
- Consider the hydrotreating of this organic substance:H2NCH2CSCH2COOH +___ H2(g) → (a) Complete a balanced chemical equation to producethe most common hydrogen compounds of N, S, andO and a saturated hydrocarbon.(b) Why is more hydrotreating needed after coking thanafter hydrocracking?arrow_forwardWhy do fatty acid esters (the biodiesel) and glycerin separate into different layers?arrow_forwardExplain what property of carbon makes it essential for organic life?arrow_forward
- 1. What is formed when a halogen molecule replaces hydrogen molecule(s) in an aromatic hydrocarbon? 2. What is formed when hydrogen molecules are replaced in an aliphatic hydrocarbon by halogen molecules? 3. What are a class of chemical compounds containing a hydroxyl group (-OH) bonded directly to an aromatic hydrocarbon group (like benzene)? 4. used to test for the presence of aromatic compounds in alcohols? 5. The dehydration of alcohols in the formation of ethers happens at _________.arrow_forwardWhat characteristics of carbon make it ideal for the formation of organic compounds?arrow_forwardPolystyrene is not biodegradable. Referring to it's chemical structure (chemical bonds/functional groups etc.) explain in one line what gives it this property.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





