
Concept explainers
(a)
Interpretation: A cyclic compound that is an isomer of trans-2-butene needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(a)
Answer to Problem 49E

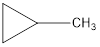
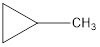
Explanation of Solution
The formula of trans-2-butane is C4H8. The cyclic compounds that is an isomer of trans-2-butene are as follows:

(b)
Interpretation: An ester that is an isomer of propanoic acid needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(b)
Answer to Problem 49E
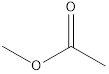
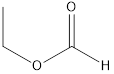
Methyl ethanoate ethyl methanoate
Explanation of Solution
The molecular formula of propanoic acid is
Esters which are isomers of propanoic acid are as follows:
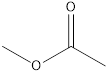
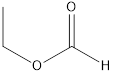
Methyl ethanoate ethyl methanoate
(c)
Interpretation: A
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(c)
Answer to Problem 49E
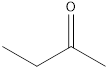
2-butanone
Explanation of Solution
The molecular formula of butanal is
A ketone that is an isomer of butanal is as follows:
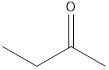
2-butanone
(d)
Interpretation: Secondary
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(d)
Answer to Problem 49E
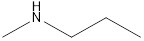

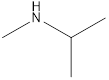
Methyl-propyl-amine Diethyl-amine Isopropyl-methyl-amine
Explanation of Solution
Secondary amine that is an isomer of butylamine are,
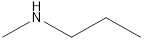

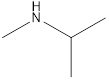
Methyl-propyl-amine Diethyl-amine Isopropyl-methyl-amine
(e)
Interpretation: A tertiary amine that is an isomer of butylamine needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(e)
Answer to Problem 49E
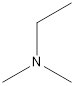
Ethyl-dimethyl-amine
Explanation of Solution
A tertiary amine that is an isomer of butylamine is as follows:
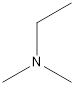
Ethyl-dimethyl-amine
(f)
Interpretation: An ether that is an isomer of 2-methyl-2-propanol needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(f)
Answer to Problem 49E
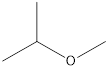


2-Methyoxy-propane Ethoxy-ethane 1-Methoxy-propane
Explanation of Solution
The formula of 2-methyl-2-propanol is
Ethers that are isomers of 2-methyl-2-propanol are as follows:
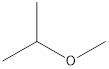


2-Methyoxy-propane Ethoxy-ethane 1-Methoxy-propane
(g)
Interpretation: A secondary alcohol that is an isomer of 2-methyl-2-propanol needs to be drawn.
Concept Introduction : Structural isomers have the same molecular formula. The bonding pattern and the arrangement of the atoms are different in different isomers.
(g)
Answer to Problem 49E
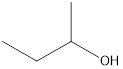
2-butanol
Explanation of Solution
The formula of 2-methyl-2-propanol is
Secondary alcohol that is an isomer of 2-methyl-2-propanol is as follows:
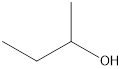
2-butanol
Want to see more full solutions like this?
Chapter 21 Solutions
CHEM.PRINC.W/OWL2+REBATE+2 SUPPL.>IP<
- Which compounds in Figure 24 are both a tertiary amine and a tertiary alcoholarrow_forwardExplain Amines ?arrow_forwardWhat are amines? What is its general formula? What are the properties of amines? What are the steps in the IUPAC and common naming of amines? Draw 5 condensed structures of amines and write their IUPAC name (do not include those in the video. What are the uses of amines? Cite examples.arrow_forward
- Which statements are true about amines? NH3+ is the form found in our blood amines can react with aldehydes to produce ethers amines act as weak acids in aqueous solutions amines can react with carboxylic acid to produce alcoholsarrow_forwardWhat kinds of functional groups are in Caffeine? I got 2 amides, one amine, and one alkene functional group.arrow_forwardWhat functional group found in kratom, ibogaine and heantosarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




