
Interpretation: Electron dot structures of Ethene and Ethyne are to be drawn. Also, describe the shape of each molecule.
Concept Introduction: Electron dot structures are also called Lewis dot structures.
Answer to Problem 18LC
Electron dot structure of Ethene Electron dot structure of Ethyne
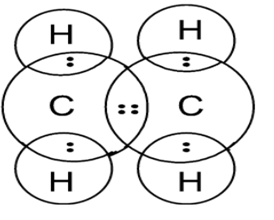
Shape of the Ethene molecule is Trigonal Planar.
Shape of the Ethylene molecule is Linear.
Explanation of Solution
Given Information: Shape of molecules Ethene and Ethyne depends upon bonding theories.
Electron dot structure of Ethene
Total number of valence electrons present are 12, the central atom is Carbon and Hydrogen is a side atom. Each Carbon has 4 valence electrons from where 2 electrons from each Carbon are used to make a double bond between them. Remaining 2 electrons of Carbon are used to make a total of 4 single bonds with 4 Hydrogen single atoms.
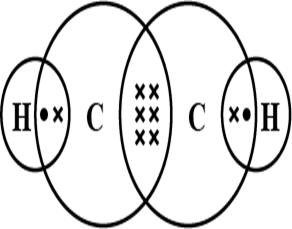
Electron dot structure of Ethyne
Total number of valence electrons present are 10, the central atom is Carbon and Hydrogen is a side atom. Each Carbon has 4 valence electrons from which 3 electrons from each Carbon is used to make a triple bond between them. Remaining 1 electron of Carbon is used to make a total of 2 single bonds with 2 Hydrogen single atoms.
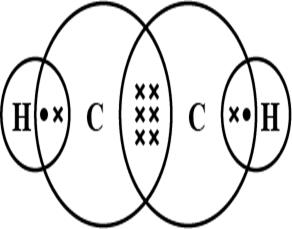
Shape of the Ethene molecule is Trigonal Planar as we know Ethene has a formula CH2=CH2 where each Carbon is attached to 3 atoms- 2 Hydrogen and 1 Carbon forming the Trigonal planar shape.
Shape of the Ethyne molecule is Linear as all 4 atoms lie in a straight line.
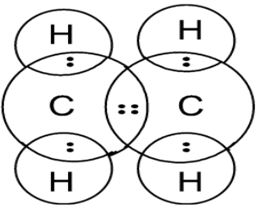
Electron dot structure of Ethene Electron dot structure of Ethyne
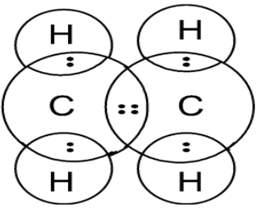
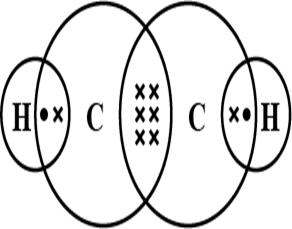
Shape of the Ethene molecule is Trigonal Planar.
Shape of the Ethylene molecule is Linear.
Chapter 22 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





