
Concept explainers
Interpretation:
The products obtained from the following reactions are to be determined: treating tyrosine with excess bromine water, reaction of phenylalanine with ethanol in the presence of hydrogen chloride, and the reaction of alanine and benzoyl chloride in aqueous base.
Concept introduction:
Amino acids contain both carboxyl group and an amino group. Both, carboxyl groups and amino groups donate and accept protons from water, respectively, to form ions. Amino acids form double ionized species by this exchange of protons in a solution between carboxyl group and amino group. These double ionized species are termed as zwitter ions.
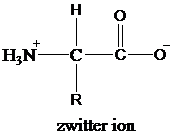
Deactivating groups like
Amino acids reacts with alcohols to produce esters, and follow nucleophilic aryl substitution reaction and the process is called Fischer esterification.
Amino acids react with acid chlorides to form acetylation or benzoylation of amino acids occur.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Organic Chemistry
- Somatostatin is a tetradecapeptide of the hypothalamus that inhibits the release of pituitary growth hormone. Its amino acid sequence has been determined by a combination of Edman degradations and enzymic hydrolysis experiments. On the basis of the following data, deduce the primary structure of somatostatin: 1. Edman degradation gave PTH-Ala. 2. Selective hydrolysis gave peptides having the following indicated sequences: Phe-Trp Thr-Ser-Cys Lys-Thr-Phe Thr-Phe-Thr-Ser-Cys Asn-Phe-Phe-Trp-Lys Ala-Gly-Cys-Lys-Asn-Phe 3. Somatostatin has a disulfide bridge.arrow_forwardDraw the structure of the tetrapeptide Asp-Arg-Val-Tyr. Please show the appropriate stereochemistry of the natural amino acids in the resulting peptide. Please draw all ionizable groups in their neutral form.arrow_forwardThe peptide Proline-Serine-Alanine-Phenylalanine-Glutamine is present at pH 7. Draw the peptide and include stereochemistry.arrow_forward
- Write a structural formula for the product formed by treatment of the N-terminal amino group with Sanger’s reagent and propose a mechanism for its formation.arrow_forward4b) Canavanine is closely related to arginine, and like arginine its side group has a +1 charge when protonated. If you dissolved canavanine in an aqueous solution at pH 10, what would the net charge on a molecule of canavanine be? Please show your work or make it clear how you determined the charge contribution from each ionizable group.arrow_forwardTetrapeptide A is completely hydrolysed under acidic conditions to yield alanine, aspartic acid, isoleucine and valine. On neutralisation of the hydrolysate, an odour of ammonia is detected. The reaction of the tetrapeptide with phenylisothiocyanate, followed by mild hydrolysis yields no phenylthiohydantoin derivative. Incubation of A with carboxypeptidase A has no effect either. Explain...arrow_forward
- A tripeptide on hydrolysis produced glycine, alanine and leucine. The structures of these amino acids are shown below. On reaction with Edman’s reagent, leucine was released as the phenylhydantoin. Treatment of the tripeptide with carboxypeptidase gave glycine. Draw the structure of the tripeptide.arrow_forwardBrucine is a poisonous alkaloid obtained from Strychnos nux vomica, a tree that grows in India, Sri Lanka, and northern Australia. Write out a resolution scheme, which shows how a racemic mixture of phenylalanine can be resolved using brucine.arrow_forwardAn unknown decapeptide was isolated and characterized. Complete hydrolysis of this peptide gave : F(2), A,G,C,K,N,T, W and V. Treatment with carboxypeptidase releases A. Reaction with Edman’s reagent gave PTH-T and a nonapeptide. The nonapeptide was treated with trypsin and gave 2 peptides: (V-C-G-A) and (N-FF-W-K). Give the sequence of amino acid in the decapeptide.arrow_forward
- Suggest a scheme you will follow to synthesise the dipeptide Ala-Glyarrow_forwardSuggest how you would separate the free l-amino acid from its acylated d enantiomerarrow_forwardAnother strategy used to resolve amino acids involves converting the carboxy group to an ester and then using a chiral carboxylic acid to carry out an acid–base reaction at the free amino group. Using a racemic mixture of alanine enantiomers and (R)-mandelic acid as resolving agent, write out the steps showing how a resolution process would occur.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





