
Concept explainers
(a)
Interpretation:
The type of the following organic reaction should be identified.
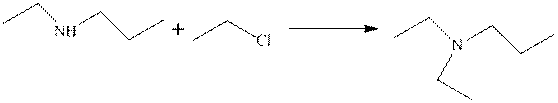
Concept introduction:
Nucleophilic substitution reaction is an organic reaction that includes the attack of a nucleophile on electrophilic center along with the removal of the leaving group.
(b)
Interpretation:
The type of the following organic reaction should be identified.
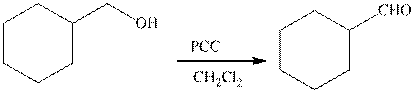
Concept introduction:
Oxidation reaction is defined as the addition of oxygen atom to the reactant molecule that causes the increase of the oxidation state of the reactant molecule.
Pyridinium chlorochromate(PCC) is a derivative of chromic acid but it is milder in nature. It is employed in the oxidation reaction of primary and secondary alcohol. The primary alcohol is oxidized to form
(c)
Interpretation:
The type of the following organic reaction should be identified.
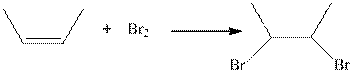
Concept introduction:
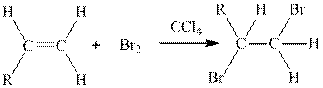
Where, R is an alkyl group.
(d)
Interpretation:
The type of following organic reaction should be identified.
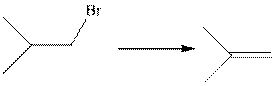
Concept introduction:
Elimination reaction is the type of reaction in which two substituents are removed from the reactant molecule to form the product Elimination reaction is of two types:
1.
2.
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
GENERAL CHEMISTRY(LL)-W/MASTERINGCHEM.
- Shown below is a carbocation intermediate in an electrophilic addition reaction of HCl with two different alkenes. Draw structural formulas for both of the alkenes.arrow_forwardIf possible please provide the products produced from the following reactions.arrow_forwardIn the reaction of ethylene with H20O in the presence of sulfuric acid,which one adds across the double bond first? *arrow_forward
- How is alkene produced by Kolbe’s electrolytic method?arrow_forwardHow much free energy is released under standard conditions when electrons are passed from succinate to ubiquinone? Note: The Faraday constant F = 23.1 kcal/(mole x volt) A) About 0.069 kcal/mol B) About 54 kcal/mol C) About 35 kcal/mol D) About 1.6 kcal/mol E) None of the above, energy is required for electrons to flow from succinate to ubiquinonearrow_forwardDraw the structure of the major organic product you would expect from the reaction of 1-bromopropane with Nal. You do not have to explicitly draw H atoms. • Draw only the product derived from 1-bromopropane. Do not draw other organic by-products.arrow_forward
- When ClO3- is oxidized, a possible product isarrow_forwardII. Synthesis using Condensation Reactions Complete the following syntheses starting with one of the condensation reactions we have learned and any other necessary reactions. 1) orb suivong bre tono -COOH 4 25 sohas potentialarrow_forwarde discuss SN1arrow_forward
- 5. Complete the missing reactant and identify the type of reaction CaCO3-CaO+? a) b) ?? + 2AgNO3-2AgCl+Ca(NO3)2 c) ?? + H2SO4- BaSO4+2H++2Cl-.arrow_forwardWe have a hydrocarbon, C16H26. The compound has two triple bonds and the acids produced by ozonolysis are CH3(CH2)4CO2H and H2OCCH2CO2H. What is a likely structure for this hydrocarbon?arrow_forward13 Write and name the other product of each of the following reactions: Acid catalyst C4H9OH …………………. + H2O (Concentrated Sulphuric acid) Acid catalyst C3H7OH …………………. + H2O (Concentrated Sulphuric acid)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





