
Concept explainers
(a)
Interpretation: Dotted lines to show
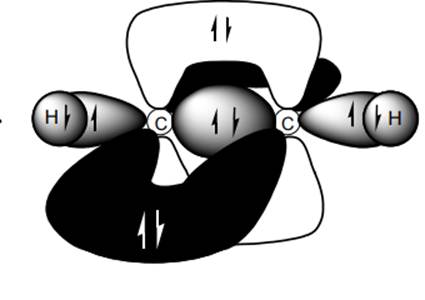
Concept introduction: Bonds formed due to head-to-head overlap are termed sigma bond while ones formed by sideways or lateral overlap are named pi-bonds.
A single bond has one
(b)
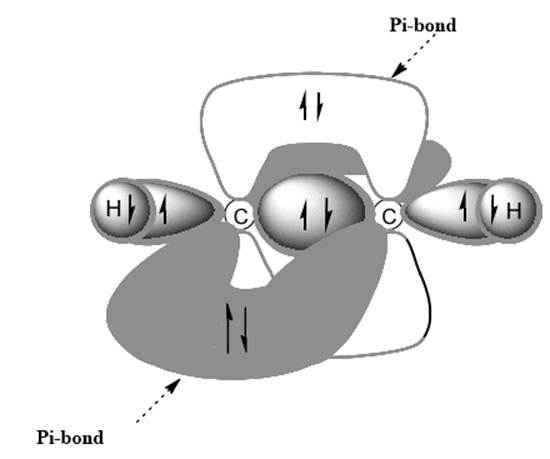
Interpretation: Dotted lines to show
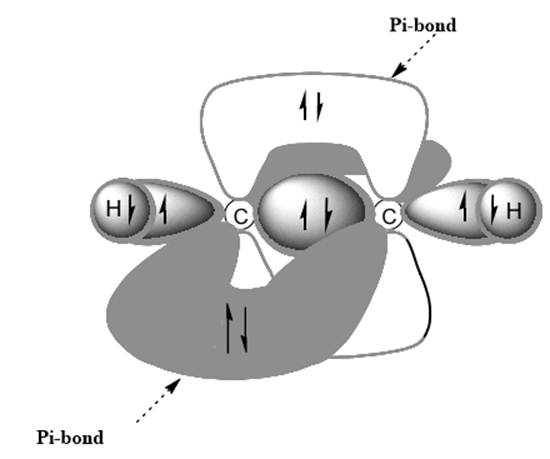
Concept introduction: Bonds formed due to head-to-head overlap are termed sigma bond while ones formed by sideways or lateral overlap are named pi-bonds.
A single bond has one

Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: A Guided Inquiry
- i think E is the right molecule what about you ??arrow_forward2. Use the curved arrow formation to show how the electrons flow in the distance from on the left to give the one on the rightarrow_forward3) Combine any Atomic orbitals to form Molecular orbitals assuming they fulfill the phase and energy requirement. Construct MO diagram (include AOs, MOs, pictures of each, electrons, HOMO, LUMO, B.O., and comparative energies, etc.). REMOVE an ELECTRON to form CATION Please only do part 3. The first image shows the problemarrow_forward
- please do both with explanation , if u hv plan to do only 1 then please skip lets some other do allarrow_forward36a,. Solve the problem in the picture,arrow_forwardQualitatively, how will the potential energy diagram for H2 be altered for N2? Draw one qualitative figure showing the potential energy curve for H2, and then add another curve for N2. Briefly explain how and why they differ.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

