
Concept explainers
(a)
Interpretation: Whether the proposed statement by the student about the bond shown below as triple bond should be explained with appropriate argument.
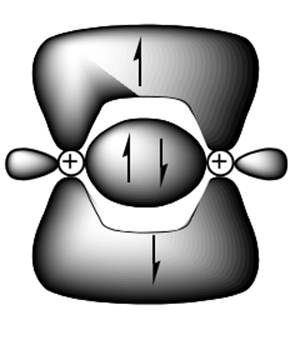
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy. This phenomenon is referred as hybridization.
A single bond has one
(b)
Interpretation: The number of electrons present in a triple bond and in one representation shown below should be indicated.
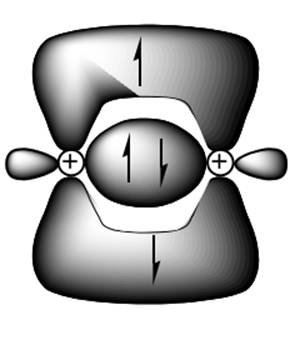
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy. This phenomenon is referred as hybridization.
A single bond has one
(c)
Interpretation: Misconception that might have led to bond identified as triple bond by the student should be known.
Concept introduction: The 2 types of overlapping are as follows:
1. Head-on overlapping (sigma bond)
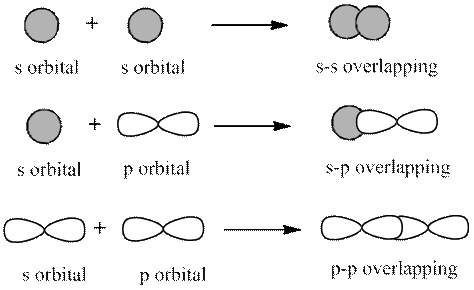
2. Sideway overlapping (pi bond)
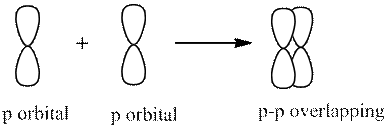
A single bond has one
(d)
Interpretation: The space that could be used to add another p bond so as to generate a triple bond should be determined.
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy although they may differ in shape and orientation. This phenomenon is referred as hybridization.
The energy and orientation of the new hybrid orbital depend upon by the kind and number of orbitals used in the hybridization. The new hybrid orbitals are always equal in number to number of atomic orbitals that combine.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: A Guided Inquiry
- Consider the incomplete orbital representation of O2 , below right. a. Identify which lobes are hybrid orbitals (identify the type) and which lobes arep orbitals. b. Use dotted lines to show any bonds. c. Use up or down arrows to show electron occupation of each hybrid orbital or bond.arrow_forwardCan I get help on the section called 'Hybrid Orbital of Central Atom' please?arrow_forwardCan you help me number 8 part i. )? Answer is false and can you explain to me?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
