
Using the
a. Determine the order of the group.
b. Verify that the E irreducible representation is orthogonal to each of the other irreduciblerepresentations.
c. For each of the irreducible representations, verify that the sum of the squares of thecharacters equals the order of the group.
d. Reduce the following representations to their component irreducible representations:
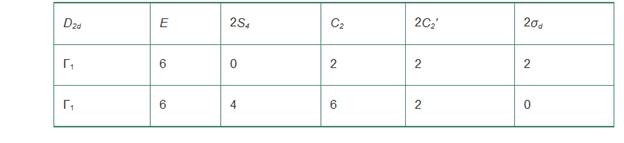
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry (3rd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Central Science (13th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
General Chemistry: Atoms First
- Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?arrow_forwardShow that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e Tdarrow_forwardShow that any irreducible representation of these point groups is normalized. a C2 b D2d c Oh d C4h e C6varrow_forward
- 4. Consider the molecule chloroform (CHCl3):Almost anything can be used to construct matrix representatives! Use just thethree Cl atoms as basis set members to derive its characters for the reduciblerepresentation and find out to which irreducible representations it can be reduced.arrow_forwardNO3- (d3h) MO diagram.. needed in irreducible formarrow_forwardD10) Determine the Irreducible representation of Tetrachloro ethene (Point group: D2h)arrow_forward
- Define the following terms: character, representation, reducible representation, irreducible representation, character tablearrow_forwardA set of basis functions is found to span a reducible representation of the group D2 with characters 6,−2,0,0 (in the order of operations in the character table in the Resource section). What irreducible representations does it span?arrow_forwardMolecule: [W(CN)8] 2- . Point Groupd D4h. Use the CN bond vectors as your basis to determine a irreducible representations and total representation.arrow_forward
- 4 Considering the carbonate atomic (CO32- ) construct the SALCS. 1) Make a table that has each atomic orbital and the corresponding symmetry operations. 2) Operate on each individual atomic orbital 3) Make the irreducible representations of the point group.arrow_forwardIrreducible representation for cH2cl2arrow_forwardThe molecule of interest PtCl4^2-. Find the irreducible representation(s) of the pz orbitals, using the pz orbitals of Cl. Also, use the projection operator method and show what one irreducible representation looks like. What orbitals if any match up with the orbital on Pt (from a pure symmetry approach)arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
