
Concept explainers
White elemental phosphorus consists of tetrahedral
a. The Raman spectrum of
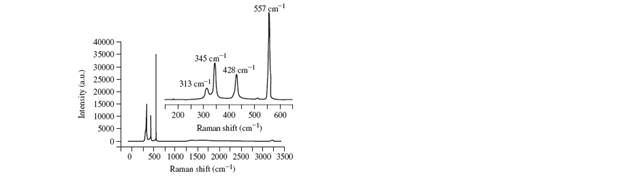
b. If
c. Could a pure sample of
Learn your wayIncludes step-by-step video

Chapter 4 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: A Molecular Approach (4th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: A Molecular Approach
General Chemistry: Atoms First
- The first LEDs were made from GaAs, which has a bandgap of 1.43 eV. What wavelength of light would be emittedfrom an LED made from GaAs? What region of theelectromagnetic spectrum does this light correspond to: ultraviolet,visible, or infrared?arrow_forwardWhat is the trend in the size of the bandgap as you move down the column of the group 4A elements?arrow_forwardSketch a tetragonal unit cell, and within that cell indicate locations of the 1 1 1/2 and point coordinates 1/2 1/4 1/2arrow_forward
- What is the minimum number of atoms that could be containedin the unit cell of an element with a face-centered cubiclattice? (a) 1, (b) 2, (c) 3, (d) 4, (e) 5.arrow_forwardShow that the radius of the largest sphere that can be placed in a tetrahedral interstitial site in an fcc lattice is 0.225r1, where r1 is the radius of the atoms making up the lattice. (Hint: Consider a cube with the centers of fourspheres placed at alternate corners, and visualize the tetrahedral site at the center of the cube. What is the relationship between r1 and the length of a diagonal of a face? The length of a body diagonal?)arrow_forwardThe unit cells of SbCl3 are orthorhombic with dimensions a = 812 pm, b = 947 pm, and c = 637 pm. Calculate the spacing, d, of the {321} planes.arrow_forward
- How many bonds does each carbonatom in C60 make? Based on thisobservation, would you expect thebonding in C60 to be more like thatin diamond or that in graphite?arrow_forwardIn a certain unit cell. planes cut through the crystal axesat (2a. 3b. c). (a. b. c). (6a. 3b. 3c). (2a. -3b. -3c). Identify theM iller indices of the planes.arrow_forwardThe equilibrium density of vacancies ns is given by Nexp(-Es/kT) , where N is the density of semiconductor atoms and Es is the energy of formation. Calculate ns in silicon at 27° C, 900 ° C, and 1200° C. Assume Es = 2.3 eV.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




