
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.39SP
Treatment of tert-butyl alcohol with concentrated HCI gives tert-butyl chloride.
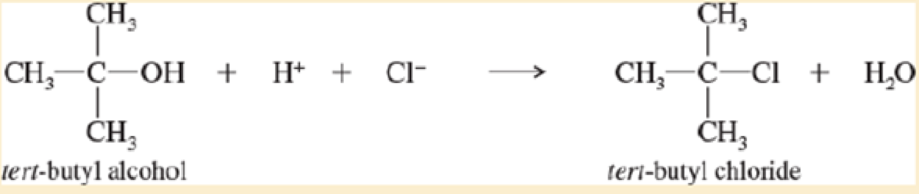
When the concentration of H+ is doubled, the reaction rate doubles. When the concentration of tert-butyl alcohol is tripled, the reaction rate triples. When the chloride ion concentration is quadrupled, however, the reaction rate is unchanged. Write the rate equation for this reaction.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify by describing the structure of the products that will form when acid-catalyzed water is removed from trans-2-methyl cyclohexanol and sodium methoxide base and hydrogen bromide are removed from trans-1-bromo 4-methyl cyclohexane.
In the reaction of acetone with sodium borohydride, what is the geometry of the reaction intermediate that results from the nucleophilic addition of the hydride ion to the carbonyl group?
A) trigonal planar B) tetrahedral
C) linear D) octahedral
Consider the reaction of NaCN with 1-bromohexane in DMSO. Predict the change in rate of reaction if the concentration of the NaCN is tripled.
A) The reaction rate stays the same
B) The reaction rate doubles
C) The reaction rate is halved
D) The reaction rate quadruples
E) The reaction rate triples
Chapter 4 Solutions
Organic Chemistry (9th Edition)
Ch. 4.3A - Draw Lewis structures for the following free...Ch. 4.3B - a. Write the propagation steps leading to the...Ch. 4.3C - Prob. 4.3PCh. 4.3C - Prob. 4.4PCh. 4.4 - The following reaction has a value of G =...Ch. 4.4 - Under base-catalyzed conditions two molecules of...Ch. 4.5B - When ethene is mixed with hydrogen in the presence...Ch. 4.5B - For each reaction, estimate whether S for the...Ch. 4.7 - a. Propose a mechanism for the free radical...Ch. 4.7 - a. Using bond-dissociation enthalpies from...
Ch. 4.8 - The reaction of tert-butyl chloride with methanol...Ch. 4.8 - Under certain conditions, the bromination of...Ch. 4.8 - When a small piece of plat num is added to a...Ch. 4.10 - Prob. 4.14PCh. 4.10 - Prob. 4.15PCh. 4.12 - The bromination of methane proceeds through the...Ch. 4.12 - a. Using me BDEs in Table4-2 (page 167 ), compute...Ch. 4.13A - What would be the product ratio in the...Ch. 4.13A - Classify each hydrogen atom in the following...Ch. 4.13B - Use the bond-dissociation enthalpies in Tabte4-2...Ch. 4.13B - Prob. 4.21PCh. 4.13B - Prob. 4.22PCh. 4.14 - a. Compute the heats of reaction for abstraction...Ch. 4.14 - 2,3-Dimethylbutane reacts with bromine in the...Ch. 4.14 - Prob. 4.25PCh. 4.15 - Prob. 4.26PCh. 4.15 - Prob. 4.27PCh. 4.16A - Prob. 4.28PCh. 4.16A - Prob. 4.29PCh. 4.16B - Prob. 4.30PCh. 4.16C - Prob. 4.31PCh. 4.16C - Acetonitrile (CH3C N) is deprotonated by very...Ch. 4.16D - Prob. 4.33PCh. 4 - The following reaction is a common synthesis used...Ch. 4 - Consider the following reaction-energy diagram. a....Ch. 4 - Draw a reaction-energy diagram for a one-step...Ch. 4 - Draw a reaction-energy diagram for a two-step...Ch. 4 - Prob. 4.38SPCh. 4 - Treatment of tert-butyl alcohol with concentrated...Ch. 4 - Label each hydrogen atom in the following...Ch. 4 - Prob. 4.41SPCh. 4 - Prob. 4.42SPCh. 4 - Prob. 4.43SPCh. 4 - Prob. 4.44SPCh. 4 - Prob. 4.45SPCh. 4 - Prob. 4.46SPCh. 4 - For each compound, predict the major product of...Ch. 4 - When exactly 1 mole of methane is mixed with...Ch. 4 - Prob. 4.49SPCh. 4 - Prob. 4.50SPCh. 4 - Prob. 4.51SPCh. 4 - When dichloromethane is treated with strong NaOH,...Ch. 4 - Prob. 4.53SPCh. 4 - When a small amount of iodine is added to a...Ch. 4 - Prob. 4.55SPCh. 4 - When healthy, Earths stratosphere contains a low...Ch. 4 - Prob. 4.57SPCh. 4 - lodination of alkanes using iodine (I2) is usually...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2,2-Dimethyl-1-propanol when introduced with Mineral Chameleon will become _______ Mineral Chameleon is an Oxidizing agent Select your answer. 2,2-Dimethylpropanal 2,2-Dimethyl-1-propene 2,2-Dimethyl-1-propanone No reaction 5-Methyl-1-octene when introduced with Water will become __________ . (Hydration Reaction) Select your answer. 5-Methyl-2-octanol 5-Methyl-1-octane 1-octene 4-Methyl-7-octanolarrow_forwardCHOOSE THE LETTER OF THE CORRECT ANSWER (the given picture is for number 1 only) 1. What structure will characterize the intermediate in this reaction mechanism, after the leaving group is removed?reaction mechanisms of organic molecules (alcohol to carboxylic acid) a. Change in conformation b. Carbocation formation c. Rearranged orbitals d. Oxonium formation 2. Based on molecular structure, alcohol molecules and ether molecules are analogues of water. This implies that both molecules can form hydrogen bonds with water molecules. Which statement is FALSE? a. Alcohol molecules are hydrogen bond donors. b. Ether molecules are hydrogen bond donors. c. Alcohol molecules are hydrogen bond acceptors. d. Ether molecules are hydrogen bond acceptors.arrow_forwardThe reaction of 1-bromopropane and sodium hydroxide in ethanol occurs by an SN2mechanism. What happens to the rate of this reaction under the following conditions?(a) The concentration of NaOH is doubled.(b) The concentrations of both NaOH and 1-bromopropane are doubled.(c) The volume of the solution in which the reaction is carried out is doubled.arrow_forward
- What reaction conditions are needed to convert (R)-2-ethyl-2- methyloxirane to (R)-2-methylbutane-1,2-diol ?arrow_forwardUsing pKa Values to Predict the Direction of Equilibrium Determine the direction of equilibrium when acetylene (HC≡CH) reacts with −NH2 in a proton transfer reaction.arrow_forwardImagine that the aldehyde 2 methylbutanal reacts with ethane-1,2-diol under acidic conditions. Draw a balanced reaction formula for the reaction and Draw the reaction mechanism for the reaction.arrow_forward
- In an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H10O) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCl) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardThe following alcohol can be formed via two different Grignard reactions. Draw the GRIGNARD REAGENT which contains a BENZENE ring. Use the covalent format R-Mg-Br instead of the ionic format. Then draw the carbonyl compound Draw the GRIGNARD REAGENT. Use the covalent format R-Mg-Br instead of the ionic format. Then draw the carbonyl compound with a benzene ringarrow_forwardGive the reagents and intermediate products for the following two-step reaction.arrow_forward
- Which of the following sets of reagents would not be an acceptable method for the preparation of ethylacetate? acetic acid, ethanol, and an acid catalyst Acetic anhydride and ethanol Sodium acetate and ethanol Sodium acetate and ethyliodidearrow_forwarddescribe how steric and electronic effects influence the postion of equilibrium when the electrophilic center of an aldehyde or ketone is under nucleophilic attack.arrow_forwardWhen 3-methyl-2-butanol is heated with concentrated HBr, a rearranged product is obtained. When 2-methyl-1-propanol reacts under the same conditions, a rearranged product is not obtained. Explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License