
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4.13B, Problem 4.20P
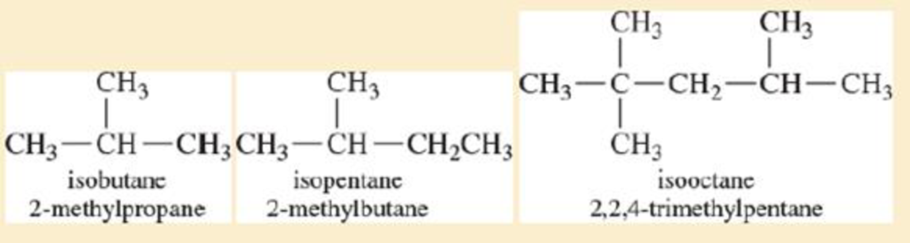
Use the bond-dissociation enthalpies in Tabte4-2 (page 167) to calculate the heats of readier for the two possible first propagation steps in the chlorination of isobutene. Use this information to draw a reaction-energy diagram like Figure4-8, comparing the activation energies for formation of the two radicals.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Sketch a potential-energy diagram for the exothermic, elementary reaction A + B→C + D
and on it denote axii labels, reactants, products, enthalpy of the reaction, and energy of activation.
Give good explanation asap
if a second half reaction has a rate of constant K equals the following
Use the bond-dissociation enthalpies to calculate the heats of reaction for the two possible first propagation steps in the chlorination of isobutane.Use this information to draw a reaction-energy diagram activation energies for formation of the two radicals
Chapter 4 Solutions
Organic Chemistry (9th Edition)
Ch. 4.3A - Draw Lewis structures for the following free...Ch. 4.3B - a. Write the propagation steps leading to the...Ch. 4.3C - Prob. 4.3PCh. 4.3C - Prob. 4.4PCh. 4.4 - The following reaction has a value of G =...Ch. 4.4 - Under base-catalyzed conditions two molecules of...Ch. 4.5B - When ethene is mixed with hydrogen in the presence...Ch. 4.5B - For each reaction, estimate whether S for the...Ch. 4.7 - a. Propose a mechanism for the free radical...Ch. 4.7 - a. Using bond-dissociation enthalpies from...
Ch. 4.8 - The reaction of tert-butyl chloride with methanol...Ch. 4.8 - Under certain conditions, the bromination of...Ch. 4.8 - When a small piece of plat num is added to a...Ch. 4.10 - Prob. 4.14PCh. 4.10 - Prob. 4.15PCh. 4.12 - The bromination of methane proceeds through the...Ch. 4.12 - a. Using me BDEs in Table4-2 (page 167 ), compute...Ch. 4.13A - What would be the product ratio in the...Ch. 4.13A - Classify each hydrogen atom in the following...Ch. 4.13B - Use the bond-dissociation enthalpies in Tabte4-2...Ch. 4.13B - Prob. 4.21PCh. 4.13B - Prob. 4.22PCh. 4.14 - a. Compute the heats of reaction for abstraction...Ch. 4.14 - 2,3-Dimethylbutane reacts with bromine in the...Ch. 4.14 - Prob. 4.25PCh. 4.15 - Prob. 4.26PCh. 4.15 - Prob. 4.27PCh. 4.16A - Prob. 4.28PCh. 4.16A - Prob. 4.29PCh. 4.16B - Prob. 4.30PCh. 4.16C - Prob. 4.31PCh. 4.16C - Acetonitrile (CH3C N) is deprotonated by very...Ch. 4.16D - Prob. 4.33PCh. 4 - The following reaction is a common synthesis used...Ch. 4 - Consider the following reaction-energy diagram. a....Ch. 4 - Draw a reaction-energy diagram for a one-step...Ch. 4 - Draw a reaction-energy diagram for a two-step...Ch. 4 - Prob. 4.38SPCh. 4 - Treatment of tert-butyl alcohol with concentrated...Ch. 4 - Label each hydrogen atom in the following...Ch. 4 - Prob. 4.41SPCh. 4 - Prob. 4.42SPCh. 4 - Prob. 4.43SPCh. 4 - Prob. 4.44SPCh. 4 - Prob. 4.45SPCh. 4 - Prob. 4.46SPCh. 4 - For each compound, predict the major product of...Ch. 4 - When exactly 1 mole of methane is mixed with...Ch. 4 - Prob. 4.49SPCh. 4 - Prob. 4.50SPCh. 4 - Prob. 4.51SPCh. 4 - When dichloromethane is treated with strong NaOH,...Ch. 4 - Prob. 4.53SPCh. 4 - When a small amount of iodine is added to a...Ch. 4 - Prob. 4.55SPCh. 4 - When healthy, Earths stratosphere contains a low...Ch. 4 - Prob. 4.57SPCh. 4 - lodination of alkanes using iodine (I2) is usually...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Describe how graphical methods can be used to determine the activation energy of a reaction from a series of data that includes the rate of reaction at varying temperatures.arrow_forwardFor the following two reactions H+Cl2HCl+Cl H+Br2HBr+Br the first reaction has a lower value of A than the second reaction. What can one say about the relative properties of the intermediates HCl2 and HBr2, just from the relative values of A?arrow_forwardBelow is a proposed mechanism for ClO–(aq) reacting with I–(aq) in the presence of OH–(aq). ClO- +H2O --> HOCl +OH- (fast, reversible)I- +HOCl --> HOI +Cl- (slow)HOI + HO- --> IO- +H2O You are trying to show that the rate law for this proposed mechanism matches an experimentallydetermined one. What is the expression for koverall or k’ for the proposed mechanism?Note: When writing an expression, H2O is not involved (just don’t include it).arrow_forward
- Overall Reaction: 2 NOBr (g) ---> 2 NO (g) + Br2 (g) Determine rate constant of reaction using the tablearrow_forwardPredict the products the reactions, if there is none, write NO RXN. Also indicate, if the reaction isfast or slow.arrow_forwardFor a certain alkane the activation energies for the rate determining step of fluorination and chlorination are 8.0 kJ/mole and 20 kJ/mole, respectively. Calculate the ratio of the rate of fluorination to that of chlorination at 27oC assuming that the values of A (Arrhenius constant) are the same. (R=8.314 J/mol-K)arrow_forward
- Define the following :(i) Elementary step in a reaction(ii) Rate of a reactionarrow_forwardA reaction is found to undergo a multi step mechanism, with the steps outlined below step 1 E+D --------2C+B step 2 B+2D-----C+E -the intermediate reaction is : ?.The catalysts in this reaction is :?. The overall reaction is :?---------?.arrow_forwardAssume the reaction: CH3CH=CHCOONa + H2 + Ni ----> ??? a. Determine the correct rate law expression based on the elementary reaction. b. Draw the structure of the transition state. c. Draw the reaction coordinate diagrams for: the reaction with the catalyst and the reaction without the catalyst, but adjacent graphs. (Assume that the reaction is exothermic and has 1 transition state.) d. Does heat increase the rate of the forward reaction in the given? Why or why not?arrow_forward
- f a reaction has a rate constant of 3.7 x 10-3 s-1 at 25 oC, and an activation energy of 43.6kJ/mol. What will be the rate constant for this reaction at 75 oC?arrow_forwardKinetics is the study of reaction rates (i.e. how fast or slow a reaction proceeds). Describe 3 ways that the rate of a reaction could be increased.arrow_forwardA certain reacion proceeds three times faster at 585K than it did at 535K. Calculate the activation energy in kJ/mol for this reactionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY