
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 21PP
Practice Problem 5.21
The following are formulas for three compounds, written in noneclipsed conformations. In each instance tell which compound (A, 8, or C on the previous page) each formula represents.
(a)
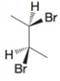
(b)
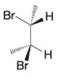
(c)
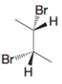
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the destabilization present in each eclipsed conformation.
Provide the least stable conformation of the following compound. I
How the relative stability of the two conformations of any disubstituted cyclohexane can be analyzed ?
Chapter 5 Solutions
Organic Chemistry
Ch. 5 - Prob. 1PPCh. 5 - Prob. 2PPCh. 5 - Prob. 3PPCh. 5 - Prob. 4PPCh. 5 - Prob. 5PPCh. 5 - Prob. 6PPCh. 5 - Prob. 7PPCh. 5 - Practice Problem 5.8 Write three-dimensional...Ch. 5 - Prob. 9PPCh. 5 - Prob. 10PP
Ch. 5 - Practice Problem 5.11 List the substituents in...Ch. 5 - Prob. 12PPCh. 5 - Practice Problem 5.13 Tell whether the two...Ch. 5 - Prob. 14PPCh. 5 - Prob. 15PPCh. 5 - Prob. 16PPCh. 5 - Prob. 17PPCh. 5 - Prob. 18PPCh. 5 - Prob. 19PPCh. 5 - Prob. 20PPCh. 5 - Practice Problem 5.21 The following are formulas...Ch. 5 - Practice Problem 5.22 Write three-dimensional...Ch. 5 - Prob. 23PPCh. 5 - Practice Problem 5.24 Give names chat include (R)...Ch. 5 - Prob. 25PPCh. 5 - Prob. 26PPCh. 5 - Prob. 27PPCh. 5 - Prob. 28PPCh. 5 - Practice Problem 5.29 Write formulas for all of...Ch. 5 - Prob. 30PPCh. 5 - Prob. 31PPCh. 5 - Prob. 32PPCh. 5 - Prob. 33PCh. 5 - Prob. 34PCh. 5 - 5.35 Designate the (R) or (S) configuration at...Ch. 5 - Prob. 36PCh. 5 - (a) Write the structure of...Ch. 5 - Shown below are Newman projection formulas for...Ch. 5 - 5.39 Write appropriate structural formulas...Ch. 5 - Discuss whether each of the following compounds...Ch. 5 - Prob. 42PCh. 5 - Prob. 43PCh. 5 - Compound F has the molecular formula C5H8 and is...Ch. 5 - Prob. 45PCh. 5 - Prob. 46PCh. 5 - Prob. 47PCh. 5 - For the following molecule, draw its enantiomer as...Ch. 5 - 5.49 (Use models to solve this...Ch. 5 - 5.50 (Use models to solve this...Ch. 5 - (Use models co solve this problem.) Write...Ch. 5 - 5.52 Tartaric acid was an important compound in...Ch. 5 - Prob. 53PCh. 5 - Prob. 54PCh. 5 - Prob. 55PCh. 5 - Prob. 1LGPCh. 5 - Prob. 2LGPCh. 5 - Prob. 3LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
For the generic equilibrium HA(aq) ⇌ H + (aq) + A- (aq), which of these statements is true?
The equilibrium con...
Chemistry: The Central Science (13th Edition)
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Q3. Which change is a physical change?
a) Wood burning
b) Iron rusting
c) Dynamite exploding
d) Gasoline ev...
Chemistry: A Molecular Approach (4th Edition)
Ortho-phosphoric acid (H3PO4) is produced as a dilute aqueous solution that must be concentrated before further...
Elementary Principles of Chemical Processes, Binder Ready Version
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- According to the following structure, draw the Newmann projections in which you must indicate: a) the most stable conformation and explain why it is more stable, b) the least stable conformation and explain why; c) a conformation where there is a gauche interaction.arrow_forwardProvide a Newman projection of a staggered conformation along the C2-C3 bond of the structure below.arrow_forwardFor the following compound - calculate the degrees of unsaturation and enter that number into the space provided.show the completed mathematical calculation. Figures form Academia Obscura (anthonycrasto.wordpress.com)arrow_forward
- Give a clear handwritten answer with explanation...give a conformer of the given molecule using line angle drawing, if possible. If not possible explain why in space provided below...?arrow_forwardBetween the cis and trans configuration of 1-chloro-4-methylcyclohexane, which is the most stable stereoisomer and configuration? To prove your argument, draw a model or use a modeling software to model cis- and trans-1-chloro-4-methylcyclohexane (show all the hydrogens and ensure proper chair conformation) and use arrows to show where steric strain occurs.arrow_forwardThe following are representations of two forms of glucose. The six-membered ring is known to exist in a chair conformation in each form. Draw clear representations of the most stable conformation of each. Are they two different conformations of the same molecule, or are they stereoisomers that cannot be interconverted by rotation about single bonds? Which substituents (if any) occupy axial sites?arrow_forward
- Draw each of the above molecules in a 3D perspective. Show all six-membered rings as chairs and all acyclic torsions in staggered conformation. If multiple conformations are possible, choose the best one.arrow_forwardImagine that this worksheet has sucked you the plane of the page, and you are now facing the indicated bond of the drawn molecule. a. Draw the six main Newman projections for the indicated bond: three eclipsed, three staggered. b. Label which conformers are eclipsed ands taggered.c. Identify the most and least stable conformer, and explain your reasoning.arrow_forwardExplain briefly and clearly the following concepts, taking as reference the molecule of n-butane and the corresponding drawings or illustrations. See pages 149-152 of the book Organic Chemistry, sixth edition (J. G. Smith). 4. What is steric hindrance in a conformation? Then draw a picture to illustrate the concept? 5. What is the torsional stress of a conformation? Then draw a picture to illustrate the concept? 6. Describe 1,3-diaxial interaction and illustrate with a specific example.arrow_forward
- Draw the Newmann projections of this molecule along the circled bond of this molecule, from the point of view of the eye. You should include and label the syn-periplanar, gauche, anticlinal and anti-periplanar conformations, and whether the conformation is staggered or eclipsed. List these four conformations from most stable to least stable?arrow_forwardIn the space provided below, write a line structure corresponding to the systematic name (R)-3-methylhexane. Then, provide a Newman projection that illustrates the highest energy conformation of (R)-3-methylhexane along the C3-C4 bond axis of the same. Again, place your answer in the space provided.arrow_forwardConsider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest energy (b) The staggered conformation(s) of highest energyarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License