
Interpretation:
Four isomeric methyl esters that have six carbon atoms and saturated carbon chain has to be given with their IUPAC names.
Concept Introduction:
Organic compounds are represented shortly by the molecular formula and structural formula. Each and every compound has its own molecular formula. Compounds can have same molecular formula but not same structural formula.
Isomers are the compounds that have same molecular formula but different structural formula. The main difference lies in the way the atoms are arranged in the structure. Isomers have different chemical and physical properties even when they have same molecular formula.
For naming an ester, it can be structurally viewed in a way that contains an acyl group and an alkyl group.
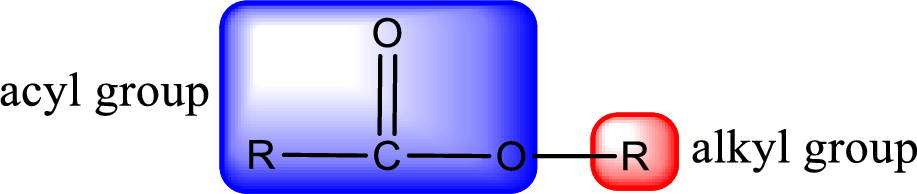
Rules to obtain IUPAC name and common name for an ester:
- Alkyl part appears first in the IUPAC name and it is followed by the acyl part of ester as a separate word.
- Name of the alkyl part in the ester is just a name of R group. It can be alkyl, cycloalkyl, or aryl group.
- Acyl part present in the ester is named by considering the acid name and replacing the suffix “-ic acid” with “-ate”.
- To obtain the common name the alkyl part name is the same while the acyl part name is derived from the common name of the acid by replacing the suffix “-ic acid” with “-ate”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic And Biological Chemistry
- provide the IUPAC name of the product when 2-butanol undergoes oxidationarrow_forwardGive the IUPAC names of structures containing two carbon atoms for the following classes of compounds: (a) ether: (b) aldehyde: (c) carboxylic acid: (d) ester:arrow_forwardGive the IUPAC name for the following compound. a) ethyl propanoate b) ethyl propyl ester c) ethyl propyl ether d) propyl ethanoatearrow_forward
- Write an equation representing all reactants and products as condensed structural formulas Dehydration of 3-methyl-2-heptanol Esterification of 1-butanol and propanoic acid Acid hydrolysis of methyl pentanoate Oxidation of 2-pentanolarrow_forwardWrite two complete balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.(a) propanol is converted to dipropyl ether(b) propene is treated with water in dilute acid.arrow_forwardIdentify the carboxylic acid derivative present and then give the IUPAC name of the compound:arrow_forward
- Write an equation representing all reactants and products as condensed structural formulas. Be sure to include any catalysts used in the reactions. Give the IUPAC name of the product(s). Dehydration of 3-methyl-2-heptanol Esterification of 1-butanol and propanoic acid Acid hydrolysis of methyl pentanoate Oxidation of 2-pentanolarrow_forwardGive the IUPAC name of the following aldehydes and ketones.arrow_forwardComplete the following chemical reaction by showing the structural formulas of all products The hydration of 1-pentene Thank you!arrow_forward
- Give the IUPAC name of the following compounds, consider alcohol and phenol as the primaryarrow_forwardDraw the structural formula and give the IUPAC name for the ester that results when 3-chlorohexanoic acid reacts with phenol.arrow_forwardWhat are the procedures to prepare aldehydes and ketones? List the reactions and include the reagents and catalyst involved.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

