
Concept explainers
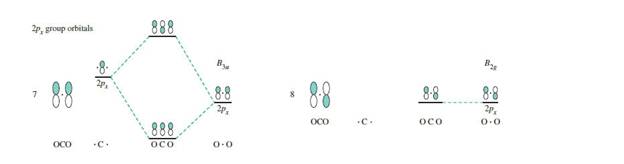
Using orbital potential energies, show that group orbital 4 is more likely than group orbital 2 to interactstrongly with the
The
orbitals of oxygen. Therefore, group orbital 6 is nonbonding.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Inorganic Chemistry
- The molecule PtCl 2 - is an important square planar starting material in synthetic inorganic chemistry. Using the p orbitals of Cl as the basis find the irreducible representations of the p orbitals and indicate which orbitals could possibly overlap with any of the orbitals on Pt . Assume that the pz orbitals on Cl are orientated in the same direction as the principle rotation axis, px orbitals are pointed at the Pt , and the py orbitals are orthogonal to the px orbitals. Show the characters of each orbital and identify which orbitals on Pt would match from a pure symmetry standpoint.arrow_forwardStudy MFT370, Fig.10.8. Infer the molecular orbital energy level diagram of the CN- ion by sketching the overlap of all the relevant atomic orbitals.arrow_forwardAssuming the polyatomic molecule SF6 and it’s cationic specie SF6+ were given, use molecular orbital theory to establish their ground state electronic configurationarrow_forward
- By using FMO (orbital coefficients and delta EHOMO-LUMO), ALSO sizes of molecules, explain the regioselectivity .arrow_forwardAssign the following molecules to point groups: (i) HF, (ii) IF7 (pentagonal bipyramid), (iii) ClF3 (T-shaped), (iv) Fe2(CO)9, (v) cubane, C8H8, (vi) tetrafluorocubane, C8H4F4arrow_forwardDraw the molecular orbital energy diagram (like we did in class, not the fancy general chemistry diagrams) for the bond between the nitrogen atom labeled a and the carbon atom labeled b in the following molecule. Clearly indicate which atomic orbitals are interacting, which resulting orbital the electrons are in, and label HOMO, LUMO, bonding and antibonding orbitals, and sigma (o), sigma* (o*), pi (n), and pi* (7*) where appropriate.arrow_forward
- Assign the following molecules to point groups: (i) HF, (ii) IF7 (pentagonal bipyramid), (iii) CIF3 (T-shaped), (iv) Fe2(CO)9 (Image 3), (v) cubane (C8H8), (vi) tetrafluorocubane (C8H4F4) (Image 4).arrow_forwardUsing huckle's molecular orbital theory find the secular determinants and the localisation energy of the given polyenesarrow_forwardIs the Be2 dimer expected to be stable? Use molecular orbital theory to justify your argument.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




