
Interpretation:
The
Concept Introduction:
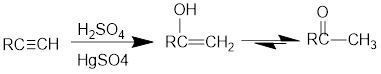
Acid Catalysed addition of water: When water is added to alkyne in the presence of an acid, the product formed will be an enol. Enol contains a double bond and
If a carbonyl group is bonded to two alkyl groups, it is called as a ketone. The enol formed in the acid catalysed addition of water will be easily converted into ketone.
Conversion of terminal
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry (3rd Edition)
- What is the major product obtained from the acid-catalyzed hydration of each of the following alkenes?arrow_forwardapredict the product of the following reactions of aldehyde and ketonearrow_forwardWhich of the following will not be the product of the reaction of an alkene with hot potassium permanganate? An aldehyde A ketone A carboxylic acid Carbon dioxidearrow_forward
- What are the major organic products obtained from the following reaction?arrow_forwardUsing cyclohexanone as the starting material, describe how each of the following compounds can be synthesized:arrow_forwardProvide the IUPAC name of the product when the aldehyde shown reacts with the indicated nucleophile in the appropriate conditions..arrow_forward
- Which of the following compounds will NOT undergo nucleophilic substitution reaction? isopropanol Benzoyl chloride propylene oxide butanonearrow_forwardIdentify all carbonyl compounds that can undergo nucleophilic addition reactions from the following:arrow_forwardWhich of the esters cannot undergo Claisen self‑condensation?arrow_forward
- What is the major organic product obtained from the following reaction?arrow_forwarddraw the organic products you would expect to isolate, from the following reactions (after hydrolysis)arrow_forwardAldehydes undergo nucleophilic addition reactions with potent nucleophiles such as water, alcohol and amino compounds. Which of the following nucleophiles will exhibit the greatest ability to attack the electrophilic carbon in the carbonyl group of ethanal? cyclohexyl alcohol para-chlorophenol isoamyl alcohol ortho-aminophenol An aromatic ring should satisfy Huckel’s rule, wherein the number of electrons participating in the cyclic conjugation should be equal to 4n + 2. Which of the following cyclic structures does NOT obey Huckel’s rule? Cyclobutadienyl dianion Tetrahydrofuran ring Cyclopropene structure Pyrimidine ring structure What is the role of H • ions in the nucleophilic substitution of alcohols usinghydrogen halides? Removal of an alkyl groupProtonation of -OH groupStabilization of carbocationActivation of oxygen radicalarrow_forward
