
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 52P
Propose a mechanism for the following reaction (show all curved arrows):
- a. Which step is the rate-determining step?
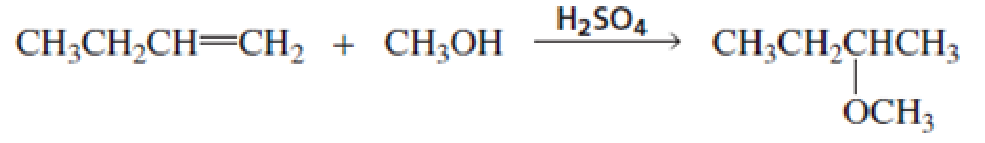
- b. What is the electrophile in the first step?
- c. What is the nucleophile in the first step?
- d. What is the electrophile in the second step?
- e. What is the nucleophile in the second step?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Whats the reason for the girgnard reagent attack that carbon of starting point in this step?
a. Bc it gives us desired product
b. Due to the sterics
c. Bc it is more electrophilic than the other carbon of the starting point
50. Why do we need to use lewis acids before electrophilic aromatic substitution reaction proceeds?
to produce a better nucleophile
to make the reaction proceed faster
to make the starting material more activated
to produce a better electrophile
I don’t get how to get the last one. I don’t use too many complicated reagents. But I have no idea how to do this. (B) only
Chapter 6 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are available for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.3 - Prob. 6PCh. 6.3 - Prob. 7PCh. 6.5 - Prob. 9PCh. 6.5 - Prob. 10PCh. 6.5 - a. What is the major product of each of the...
Ch. 6.5 - Prob. 12PCh. 6.6 - What stereoisomers are obtained from each of the...Ch. 6.6 - Prob. 14PCh. 6.8 - Prob. 15PCh. 6.10 - Name the following:Ch. 6.10 - Draw the structure for each of the following: a....Ch. 6.10 - Draw the structures for and name the seven alkynes...Ch. 6.10 - Name the following:Ch. 6.10 - Name the following:Ch. 6.11 - What hybrid orbitals are used to form the...Ch. 6.13 - Prob. 22PCh. 6.14 - Prob. 23PCh. 6.14 - Which alkyne would be the best one to use for the...Ch. 6.14 - Prob. 25PCh. 6.14 - Prob. 26PCh. 6.15 - Describe the alkyne you would start with and the...Ch. 6.15 - What are products of the following reactions?Ch. 6 - Prob. 29PCh. 6 - Prob. 30PCh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - What is each compounds systematic name?Ch. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - What reagents could be used to carry out the...Ch. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Answer Problem 42 using 2-butyne as the starting...Ch. 6 - What is each compounds systematic name?Ch. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Draw the keto tautomer for each of the following:Ch. 6 - Propose a mechanism for the following reaction...Ch. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What would be the reagent for each step?arrow_forwardWhich factors would favor an SN2 reaction? Choose one or more: A. a strong nucleophile B. a good leaving group C. a high concentration of nucleophilearrow_forwardWhat kind of reagent is KmnO4? Draw the possible reactions of this reagent with the specific compounds tested in the following reaction?arrow_forward
- Pleas explain how this process occurs. Identify SN1, SN2, E2, E1, nucleophiles and electrophiles.arrow_forwardexplain why carbonyl compounds are so attractive to both nucleophiles and electrophilesarrow_forwardThe following reaction takes place several times faster than the reaction of 2-chlorobutane with HO-: a. Explain the enhanced reaction rate.b. Explain why the OH group in the product is not bonded to the carbon that was bonded to the Cl group in the reactant.arrow_forward
- Can you explain how they got the minor product, show the movement of electrons etc.arrow_forwardWhich alkyl halide is more reactive in an SN2 reaction with a given nucleophile?arrow_forwardChoose one and draw the product using diethyl tartrate acid. What type of reaction is it? What are the key features such as a nucleophile and electrophile? Name the productarrow_forward
- Draw the starting material that required the reaction below.arrow_forwardPropose a mechanism for the following reaction (remember to use curved arrows to show the movement of electrons from the nucleophile to the electrophile):arrow_forwardWhat is the first step in an SN1 reaction? Group of answer choices Attack of the nucleophile. Loss of the leaving group. Protonation of the leaving group.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License