
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 6.54SP
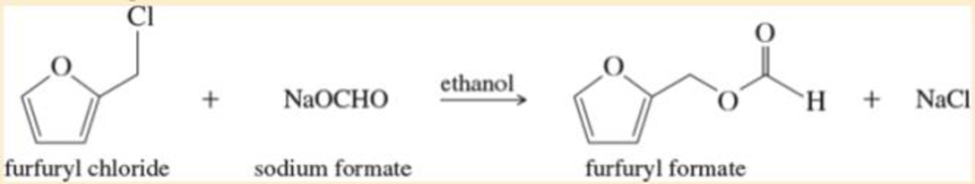
Furfuryl chloride can undergo substitution by both SN2 and SN1 mechanisms. Because it is a 1°
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The Wolff–Kishner reaction uses hydrazine (H2NNH2) and hydroxide (–OH) to reduce a carbonyl to the alkane. The first steps of the mechanism convert a carbonyl to a hydrazone in a manner similar to imine formation. Draw the mechanism arrows for the reaction from the hydrazone to the alkane. Be sure to add lone pairs of electrons and nonzero formal charges to all species.
Can you answer it and explain it? State the mechanism in which through reaction proceeds sn2 or sn1
How do we know whether a reaction occurs by the SN1 or SN2 mechanism?
Chapter 6 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 6.1 - Classify each compound as an alkyl halide, a vinyl...Ch. 6.2 - Give the structures of the following compounds. a....Ch. 6.2 - For each of the following compounds, A. give the...Ch. 6.3E - Prob. 6.4PCh. 6.4 - Prob. 6.5PCh. 6.5A - For each pair of compounds, predict which compound...Ch. 6.5B - Prob. 6.7PCh. 6.6B - Prob. 6.8PCh. 6.6B - The light-initiated reaction of...Ch. 6.6B - Show how free-radical halogenation might be used...
Ch. 6.7 - Prob. 6.11PCh. 6.7 - Prob. 6.12PCh. 6.8 - Prob. 6.13PCh. 6.9 - Predict the major products of the following...Ch. 6.9 - Prob. 6.15PCh. 6.10A - Prob. 6.16PCh. 6.11A - When diethyl ether (CH3CH2OCH2CH3) is treated with...Ch. 6.11B - Prob. 6.18PCh. 6.11B - For each pair of compounds, state which compound...Ch. 6.12 - Prob. 6.20PCh. 6.12 - Under appropriate conditions...Ch. 6.13 - Propose an SN1 mechanism for the solvolysis of...Ch. 6.13B - Prob. 6.23PCh. 6.13B - 3-Bromocyclohexene is a secondary halide, and...Ch. 6.15 - Prob. 6.25PCh. 6.15 - Prob. 6.26PCh. 6.16 - For each reaction, give the expected substitution...Ch. 6.16 - Prob. 6.28PCh. 6.16 - Prob. 6.29PCh. 6 - Prob. 6.30SPCh. 6 - Draw the structures of the following compounds. a....Ch. 6 - Give systematic (IUPAC) names for the following...Ch. 6 - Prob. 6.33SPCh. 6 - Predict the compound in each pair that will...Ch. 6 - Prob. 6.35SPCh. 6 - Give two syntheses for (CH3)2CHOCH2CH3, and...Ch. 6 - Prob. 6.37SPCh. 6 - Prob. 6.38SPCh. 6 - Chlorocyclohexane reacts with sodium cyanide...Ch. 6 - Give the substitution products expected from...Ch. 6 - Prob. 6.41SPCh. 6 - Prob. 6.42SPCh. 6 - Two of the carbocations in Problem6-42 are prone...Ch. 6 - Prob. 6.44SPCh. 6 - Predict the products of the following SN2...Ch. 6 - Prob. 6.46SPCh. 6 - Strawberry growers have used large quantities of...Ch. 6 - A solution of pure (S)-2-iodobutane ([]=+15.90) in...Ch. 6 - Prob. 6.49SPCh. 6 - Give a mechanism to explain the two products...Ch. 6 - Prob. 6.51SPCh. 6 - Because the SN1 reaction goes through a flat...Ch. 6 - Prob. 6.53SPCh. 6 - Furfuryl chloride can undergo substitution by both...Ch. 6 - Prob. 6.55SPCh. 6 - The following reaction takes place under...Ch. 6 - Propose mechanisms to account for the observed...Ch. 6 - Prob. 6.58SPCh. 6 - Prob. 6.59SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The reaction shown below proceeds by both an SN1 and an SN2 mechanism. Draw the products of each process and use mechanistic arguments to account for any differences in the products formed. Ph 'Br SH Basearrow_forwardWhat is the major substitution product for the following reaction? Show the mechanism for the reaction.arrow_forwardCan you explain which reactions they are undergoing sn1 or sn2 and state the mechanism and explain it pleasearrow_forward
- Come up with a detailed mechanism for the following cyclization that also includes all resonance strictures for any intermediates that form. Also, explain why the alkyl bromide portion of the molecule remains intact in the product even though the reaction conditions are strongly basic/nucleophilic. Br Br NaOMe Me Me Me Me MeOH MeO2C 0 °C MeO₂Carrow_forwardCompeting SN and E reactions. Predict whether the following reactions will proceed via substitution (SN1 or SN2), or elimination (E₁ or E₂), or whether the two will compete. Identify the major product.arrow_forwardAnswer the question below the reaction. ta The reaction above proceeds through which type of mechanism? SN2 SN1 E1 E2 OH + Excess NH4C1 H₂SO4 + H₂Oarrow_forward
- In the following electrophilic aromatic substitution reaction, the major product results in the substitution occurring at the 2 position. Starting from each potential carbocation intermediate shown, (a) draw the possible resonance structures and (b) explain why the major product is favored. Br 4 Br2 5 Br major product minor product + Br Br 2. LOarrow_forwardA carbamate can be prepared by treating an isocyanate with an alcohol, as shown here. This type of reaction is used to synthesize polyurethanes- polymers that have a wide variety of industrial applications, such as surface sealants, high-performance adhesives, and synthetic fibers. Propose a mechanism for this transformation. R'—ОН R. `N=C=0 R. OR' An isocyanate A carbamate (Substituted urethane) O=Carrow_forwardmethanol + CH3OH Suppose you were told that the above reaction was a substitution reaction but you were not told the mechanism. Evaluate the following categories to determine the reaction mechanism and then draw the structure of the major organic product. Type of alkyl halide: Type of nucleophile: Solvent: Is the product racemic?arrow_forward
- is this an E1 or E2 mechanism for this reaction? What is the major product and mechanism for it?arrow_forwardWhich SN1 reaction of each pair would you expect to take place more rapidly? to to (1) + H,O → + HCI or + MeOH – + HCI OMearrow_forwardThe SN1 mechanism starts with the rate-determining step which is the dissociation of the alkyl halide into a carbocation and a halide ion. The next step is the rapid reaction of the carbocation intermediate with the nucleophile; this step completes the nucleophilic substitution stage. The step that follows the nucleophilic substitution is a fast acid-base reaction. The nucleophile now acts as a base to remove the proton from the oxonium ion from the previous step, to give the observed product. Draw a curved arrow mechanism for the reaction, adding steps as necessary. Be sure to include all nonzero formal charges.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License