
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.11, Problem 21P
What hybrid orbitals are used to form the carbon-carbon
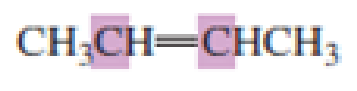


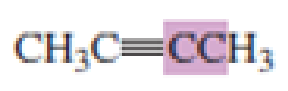

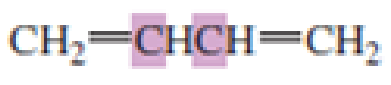

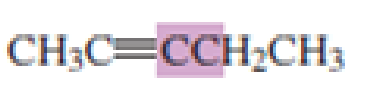
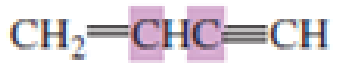
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which OH groups are bonded to sp3 hybridized carbon atoms and which are bonded to sp2 hybridized carbons?
How many un-hybridized 2p orbital (s) of the carbon atom in ethyne?
Please identify the correct ring flipped structure of compound XYZ.
Chapter 6 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are available for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.3 - Prob. 6PCh. 6.3 - Prob. 7PCh. 6.5 - Prob. 9PCh. 6.5 - Prob. 10PCh. 6.5 - a. What is the major product of each of the...
Ch. 6.5 - Prob. 12PCh. 6.6 - What stereoisomers are obtained from each of the...Ch. 6.6 - Prob. 14PCh. 6.8 - Prob. 15PCh. 6.10 - Name the following:Ch. 6.10 - Draw the structure for each of the following: a....Ch. 6.10 - Draw the structures for and name the seven alkynes...Ch. 6.10 - Name the following:Ch. 6.10 - Name the following:Ch. 6.11 - What hybrid orbitals are used to form the...Ch. 6.13 - Prob. 22PCh. 6.14 - Prob. 23PCh. 6.14 - Which alkyne would be the best one to use for the...Ch. 6.14 - Prob. 25PCh. 6.14 - Prob. 26PCh. 6.15 - Describe the alkyne you would start with and the...Ch. 6.15 - What are products of the following reactions?Ch. 6 - Prob. 29PCh. 6 - Prob. 30PCh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - What is each compounds systematic name?Ch. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - What reagents could be used to carry out the...Ch. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Answer Problem 42 using 2-butyne as the starting...Ch. 6 - What is each compounds systematic name?Ch. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Draw the keto tautomer for each of the following:Ch. 6 - Propose a mechanism for the following reaction...Ch. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following structures are identical? (Green = Cl.)arrow_forwardIn a tabular format, enumerate 5 molecules where the following atoms are found in: O (sp3-hybridized) Oxygen (sp2-hybridized) Nitrogen (sp2-hybridized) Carbon (sp2-hybridized) O (sp3-hybridized) Oxygen (sp2-hybridized) Nitrogen (sp2-hybridized) Carbon (sp2-hybridized) Hydrogen (H2o)Methanol (CH3OH)Methane (CH4)Carbon (C=C)arrow_forwardFor each highlighted carbon identify what type of orbitals it has and how many of each. Then predict the approximate bond angle about each highlighted carbon.arrow_forward
- Identify the hybridization of carbon atoms numbered 1-6 in the structure below Carbons 1 and 3 are sp3, carbons 2 and 5 are sp2, carbons 4 and 6 are sp hybridized. Carbons 2 and 3 are sp3, carbons 4 and 5 are sp2, carbons 1 and 6 are sp hybridized. Carbons 1 and 2 are sp3, carbons 3 and 6 are sp2, carbons 4 and 5 are sp hybridized. Carbons 3 and 6 are sp3, carbons 4 and 5 are sp2, carbons 1 and 2 are sp hybridized. Carbons 4 and 5 are sp3, carbons 3 and 6 are sp2, carbons 1 and 2 are sp hybridized.arrow_forwardwhat is the name of the compound? Identify the configuration of the double bond.arrow_forwardHow many chirality centers exist on this molecule? What is the absolute configuration of the primary alcohol in this structure? How many lone pairs are on the structure? How many sp2 hybridized atoms are on the structure?arrow_forward
- C2H2Br2 Describe the geometry about eachcentral carbon.arrow_forwardA.) Draw both the condensed and line-bond structure of the following compounds: 1) 3-ethylhexane 2) 1,3-dimethylcyclopentane 3) 1,3-dichloro-3-methylheptane ( Cl- , Chloro branch) 4) bromocyclobutane (Br-, Bromo branch) 5) 5-sec-butylnonane 6) 4-t-butylheptane 7) 4-ethylcycloheptene 8) 4,4-dimethylpent-2-ene 9) 3-chloro-2,7-dimethylnon-4-yne (Cl- Chloro branch) 10) 1-cyclohexylbut-2-ynearrow_forwardDraw the skeletal (line-bond) structure of (1R,2S)-1-bromo-2-methylcyclohexanearrow_forward
- Draw the energy diagram of the hybridized atomic orbitals of the oxygen atoms for a. CH3OCH2CH3 (ethyl methyl ether) b. CH3COCH3 (acetone)arrow_forwardIgnoring stereoisomers, draw the structures of the 13 constitutional isomers of molecular formula C 6H 12 that contain one carbon–carbon double bond.arrow_forward2. The carbon atom of a carbonyl group is a) sp hybridised b) sp2 hybridised c) sp3 hybridised d) none of thesearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License