
a)

Interpretation:
Electrostatic potential maps of formaldehyde is given.The nature of the formldehyde carbon atom whether electrophilic or nucleophilic is to be stated and explained.
Concept introduction:
Electrostatic potential maps are used to identify at a glance the electron rich or electron poor atoms in molecule. In them the electron rich regions are shown in red and electron poor region are shown in blue.
To state and explain:
From the electrostatic potential map of formaldehyde the nature of the formldehyde carbon atom, whether it is electrophilic or nucleophilic.
b)
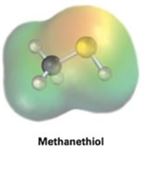
Interpretation:
Electrostatic potential map of methanethiol (CH3SH) is given. The nature of the sulfur atom in methanethiol, whether electrophilic or nucleophilic, is to be stated and explained.
Concept introduction:
Electrostatic potential maps are used to identify at a glance the electron rich or electron poor atoms in molecule. In them the electron rich regions are shown in red and electron poor region are shown in blue.
To state and explain:
From the electrostatic potential map of methanethiol, whether the sulfur atom in it is electrophilic or nucleophilic.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry
- Following is a balanced equation for bromination of toluene. (a) Using the values for bond dissociation enthalpies given in Appendix 3, calculate H0 for this reaction. (b) Propose a pair of chain propagation steps and show that they add up to the observed reaction. (c) Calculate H0 for each chain propagation step. (d) Which propagation step is rate-determining?arrow_forwardThe above reaction involves heterolytic bond breakage of HBr a) Encircle the nucleophile (s) and electrophile (s) and explain why. b) Give the mechanism of the reaction by: ) Drawing the appropriate arrows to track the flow of electrons in Showing the species formed after bond breakage and bond formation with appropriate charges. c) Predict the final products.arrow_forwardWhich is more stable - a phenyl radical, [C6H5]•, or a benzyl radical, [C6H5CH2]• - and why?arrow_forward
- Benzoic acid, Ph-COOH (C6H5CO2H), is not soluble in water while it dissolves in ether (diethyl ether), (CH3CH2)2O. Yet upon treatment with sodium hydroxide, benzoic acid turns hydrophilic and dissolves in water. Provide chemical explanation of this observation.arrow_forwardPropose a reasonable mechanism for the reaction, drawing out Lewis structure, using arrows to show electron flow, clearly identifying the bond making/bond breaking steps, and proposing reasonable intermediates for the reaction.arrow_forwardUnder milder temperatures than what is needed for dehydration, strong acids catalyze the self-condensation of 1-propyl alcohol to give di-n-propyl ether. The mechanism differs from that of today's reaction in that the protonated alcohol undergoes an Sn2 rather than an Sn1 reaction. Write the mechanism for the acid catalyzed condensation of ethyl alcohol to give diethyl ether. Make sure the mechanism includes protonation, Sn2, and deprotonation.arrow_forward
- a. Propose a mechanism for the following reaction: b. Given that ΔH° for the reaction is -42 kcal/mol and the bond dissociation enthalpies for the C¬H, C¬Cl, and O¬H bonds are 101, 85, and 105 kcal/mol respectively, calculate the bond dissociation enthalpy of the O¬Cl bond.arrow_forwardAcid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.arrow_forwardDefine the Mechanism of the Radical Addition of HBr to an Alkene ?arrow_forward
- 1-Suggest a mechanism by which the ozone, O3 is being destructed by the presence of oxygen molecule, O2, and the sun light. 2-Methyl chloride, CH3Cl, is another molecule that destructs the ozone layer. Suggest a mechanism for the destruction of ozone by methyl chloride and the sun light.arrow_forwardBromine reacts with alkenes in methanol according to the equation (see image 1). When this reaction was carried out with 4-tert-butylcyclohexene, only one isomer was formed with the molecular formula C12H23BrO (80% yield) a) Which of the following is the structure more reasonable for this compound? (see image 2) b) Explain your reasoning through a corresponding mechanismarrow_forwardcompare and construct the nucleophilic reactions of hcl with propanone, propanal and propan-1-0arrow_forward


 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


