
Concept explainers
To determine:
The formation of KCl (an ionic compound).
Introduction :
KCl or potassium chloride is an inorganic compound with a covalent bond between potassium andchloride ion. The bond is polar because chlorine is more electronegative than potassium. Acovalent bond is formed between two elements by sharing of electrons pairs to complete theirvalence shell.
Answer to Problem 12STP
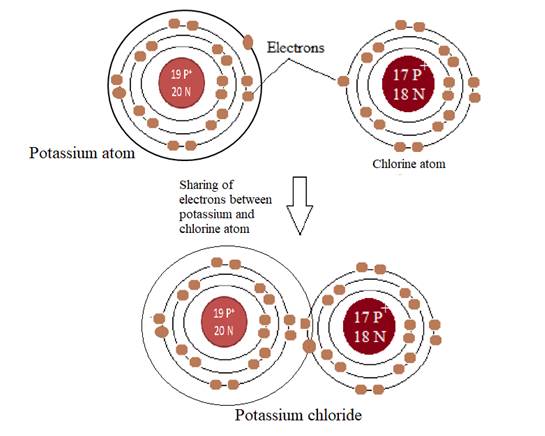
Explanation of Solution
Atoms form a bond to complete their valance shell and become stable by attaining the nearest noble gas configuration. Potassium has total 19 protons and electrons with one electron in its outer shell that can house a maximum of eight electrons to attain stability. Chlorine has atomic number 17 which suggests that the number of electrons in a chlorine atom is 17. The electronic configuration of chlorine has three shells, K, L, and M, with 7 electrons in the M shell. It needs one electron to complete its valence shell. The sharing of electrons between potassium and chlorine atom leads to a stable compound with a partial negative charge on chlorine and a partial positive charge on potassium as chlorine is more electronegative than potassium.
Chapter 7 Solutions
Biology Illinois Edition (Glencoe Science)
Additional Science Textbook Solutions
Human Anatomy & Physiology
Genetic Analysis: An Integrated Approach (2nd Edition)
Concepts of Genetics (11th Edition)
Microbiology with Diseases by Body System (4th Edition)
Biology: Life on Earth
Anatomy & Physiology (6th Edition)
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





