
Concept explainers
Interpretation:
The formation of sulfur ions needs to be explained.
Concept introduction:
The electronic configuration of atomic species is followed by set of rules defined by Aufbau’s principle, Hund’s Rule, Pauli-Exclusion principle. Each orbital is occupied by two electrons of opposite spins. As per Aufbau’s principle, electrons will occupy orbital in order of increasing energy. According to Hund’s Rule, orbitals of same energy will be filled with one electron each having the same spin.
Answer to Problem 51A
Sulfur can take up two electrons to become an octet.
Explanation of Solution
Given that the orbital notation of sulfur is given as
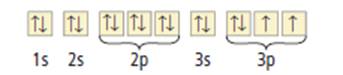
Figure 1
From the orbital notation, it can be seen that the 3p orbital has vacancy for two electrons. In other words, it can take in two electrons. To get an octet configuration, elements generally give up or take in electrons to get a stable configuration. If sulfur take up electrons, it will achieve octet configuration and become stable.
Hence, sulfur will take up two electrons to form a stable configuration.
Sulfur can take up two electrons to become an octet.
Chapter 7 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Chemistry: The Central Science (14th Edition)
Organic Chemistry
CHEMISTRY-TEXT
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





