
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 13P
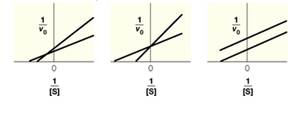
The accompanying figure shows three Lineweaver—Burk plots for enzyme reactions that have been carried out the presence, or absence, of an inhibitor. Indicate what type of inhibition is predicted based on each Lineweaver—Burk plot. For each plot indicate which line corresponds to the reaction without inhibitor and which line corresponds to the reaction with inhibitor present.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Under the following conditions, fill in the blanks. Then, describe why this inhibitor is the type of inhibitor you identified it as.
If you were to add 5nM of a reversible inhibitor, the Km for the measured enzyme catalyzed reaction would ______ (Increase, Decrease, Stay the same) to ______µM (choose appropriate value) and Vmax would _______ (Increase, Decrease, Stay the same) to ______µMs-1. So, this inhibitor is a ______ (Competitive, Uncompetitive, Mixed) inhibitor.
Conditions:
kcat = 130 s^-1
Vo = 3.0 μMs-1
S = 10 μM
Et = 0.09 µM
The rate of an enzyme-catalysed reaction was measured in the absence or presence of a non-competitive inhibitor. Which of the following Lineweaver-Burk plots would you expect to see?
a. (a)
b. (b)
c. (c)
d. (d)
Given the following data in enzyme-catalyzed reaction, what are the Vm, Km of with DEDS (presence of inhibitor) and without DEDS ( absence of inhibitor) and its type of inhibition.
Chapter 8 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 8 - Prob. 1PCh. 8 - The enzyme urease catalyzes the hydrolysis of urea...Ch. 8 - An enzyme contains an active site aspartic acid...Ch. 8 - The folding and unfolding rate constants for a...Ch. 8 - In some reactions, in which a protein molecule is...Ch. 8 - Would you expect an “enzyme” designed to bind to...Ch. 8 - The initial rate for an enzyme-catalyzed reaction...Ch. 8 - a. If the total enzyme concentration in Problem 7...Ch. 8 - Prob. 9PCh. 8 - Prob. 10P
Ch. 8 - The following data describe the catalysis of...Ch. 8 - At 37 oC, the serine protease subtilisin has kcat...Ch. 8 - The accompanying figure shows three...Ch. 8 - The steady-state kinetics of an enzyme are studied...Ch. 8 - The same enzyme as in Problem 14 is studied in the...Ch. 8 - Enalapril is an anti-hypertension “pro-drug"...Ch. 8 - Initial rate data for an enzyme that obeys...Ch. 8 - Prob. 18PCh. 8 - Suggest the effects of each of the following...Ch. 8 - The inhibitory effect of an uncompetitive...Ch. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - In kinetics experiments, the hydrolysis of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Sketch the complete reaction free energy diagram for an enzyme-catalyzed conversion of a single substrate (S) into product (P), where the reaction is spontaneous in the forward direction Overlay the free energy diagram for the uncatalyzed reaction and indicate delta delta G〒 on your sketch: Chemical step is rate limitingarrow_forwardWhat is the difference between the lock-and-key model of enzyme action and the induced-fit model?arrow_forwardGiven the following data in enzyme-catalyzed reaction, what is the Vm, Km and type of inhibition of Experiment A?arrow_forward
- The following data was obtained during kinetic analysis of an enzyme with and without an inhibitor. Substrate concentration (mM) Reaction rate without inhibitor (µM/s) Reaction rate with inhibitor (µM/s) 10 28 12 20 50 23 40 83 42 60 107 58 100 139 83 200 179 125 300 197 150 400 209 167 560 227 197 How do you calculate the KM for the enzyme in the absence of an inhibitor. And how do you calculate kcat with the given enzymatic concentration of 5 µM.arrow_forwardThe figure displays the relationship between initial rate of product formation and reactant concentration in an enzyme-catalyzed reaction with a fixed amount of enzyme. Which of the following statements best explains the shape of the rate curve at high reactant concentration?arrow_forwardDraw a hypothetical Michaelis-Menten plot for an enzyme reaction (i) without and (ii) with a pure noncompetitive inhibitor. Label Vax and Km on plotarrow_forward
- Explain why each of the following data sets from a Lineweaver–Burk plot are not individually ideal for determining KM for an enzymecatalyzed reaction that follows Michaelis–Menten kinetics.arrow_forwardIn concerted model, all subunits in the enzyme are either in the low affinity or high affinity forms.arrow_forwardExplain why the very tight binding of a substrate to an enzyme is not desirable for enzyme catalysis, whereas tight binding of the transition state is desirable.arrow_forward
- In a plot of 1/v vs 1/[S] for an enzyme-catalyzed reaction, presence of a non-competitive inhibitor will alter the: * Intercept on the 1/v axis. Intercept on the 1/[S] axis. Curvature of the plot. Kmarrow_forwardUsing the appropriate graph and table above, explain what the R48C mutation appears to be doing to the enzyme’s function. Discuss the kinetic parameter changes and their meaning in this context, not the structure of the enzyme, which was not given to you.arrow_forwardConsider the following data for an enzyme-catalyzedhydrolysis reaction in the presence and absence ofinhibitor I: Using a Michaelis-Menten plot, determine Km for theuninhibited reaction and the inhibited reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY