
Concept explainers
Materials containing the elements Y, Ba, Cu, and O that are superconductors (electrical resistance equals zero) at temperatures above that of liquid nitrogen were recently discovered. The structures of these materials are based on the perovskite structure. Were they to have the ideal perovskite structure, the superconductor would have the structure shown in pant (a) of the following figure.
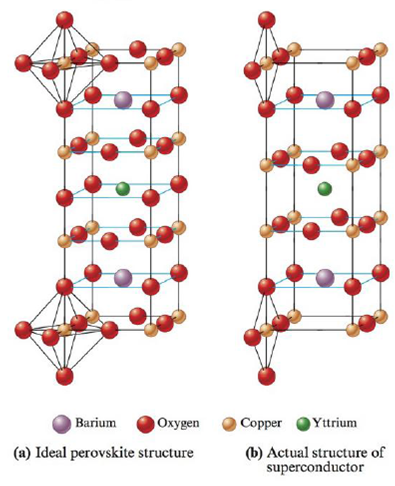
a. What is the formula of this ideal perovskite material?
b. How is this structure related to the perovskite structure shown in Exercise 85?
These materials, however, do not act as superconductors unless they are deficient in oxygen. The structure of the actual superconducting phase appears to be that shown in pan (b) of the figure.
c. What is the formula of this material?
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry: An Atoms First Approach
- In the LiCl structure shown in Figure 9.21, the chloride ions form a face-centered cubic unit cell 0.513 nm on an edge. The ionic radius of Cl- is 0.181 nm. (a) Along a cell edge, how much space is between the Cl- ions? (b) Would an Na+ ion (r=0.095nm) fit into this space? a K+ ion (r=0.133nm)?arrow_forwardA plot of In (Pvap) versus 1/T (K) is linear with a negative slope. Why is this the case?arrow_forwardRutile, TiO2, crystallizes in a structure characteristic of many other ionic compounds How many formula units of TiO2 are in the unit cell illustrated here? (The oxide ions marked by an x are wholly within the cell; the others are in the cell faces.) Unit cell for rufflearrow_forward
- The free space in a metal may be found by subtracting the volume of the atoms in a unit cell from the volume of the cell. Calculate the percentage of free space in each of the three cubic lattices if all atoms in each are of equal size and touch their nearest neighbors. Which of these structures represents the most efficient packing? That is, which packs with the least amount of unused space?arrow_forwardA metal burns in air at 600c under high pressure to form an oxide with formula MO2. This compound is 23.72% oxygen by mass. The distance between the centers of touching atoms in a cubic closest packed crystal of this metal is 269.0 pm. What is this metal? What is its density?arrow_forwardWhich solid phase that is, which allotrope of carbon is more stable, graphite or diamond? You should consult some of the tables in the thermodynamics section of this text.. Both solid phases exist under normal conditions of pressure and temperature. Explain why this is so, given that one solid phase is more thermodynamically stable than the other. Do their unit cells provide any suggestion for their relative stabilities?arrow_forward
- Outline a two-dimensional unit cell for the pattern shown here. If the black squares are labeled A and the white squares are B, what is the simplest formula for a compound based on this pattern?arrow_forwardAluminum metal crystallizes with a face-centered cubic unit cell. The volume of the cell is 0.0662 nm3. (a) What is the atomic radius of aluminum in cm? (b) What is the volume of a single aluminum atom? (c) What is the density of a single aluminum atom? (d) In face-centered cubic cell packing, the fraction of empty space is 26.0%. When this is factored in, what is the calculated density of aluminum?arrow_forwardConsider the sodium chloride unit cell shown in Figure 9.21. Looking only at the front face (five large Cl- ions, four small Na+ ions), (a) how many cubes share each of the Na+ ions in this face? (b) how many cubes share each of the Cl- ions in this face?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,





