
Concept explainers
(a)
Interpretation:
The possible resonance structures for the following skeleton structure have to be determined. Also, the resonance structure that is the most important has to be determined.
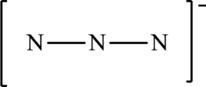
Concept Introduction:
The steps to draw the Lewis structure of the molecule are as follows:
Step 1: Find the central atom and place the other atoms around it. The atom in a compound that has the lowest group number or lowest electronegativity considered as the central atom.
Step 2: Estimate the total number of valence electrons.
Step 3: Connect the other atoms around the central atoms to the central atom with a single bond and lower the value of valence electrons by 2 of every single bond.
Step 4: Allocate the remaining electrons in pairs so that each atom can get 8 electrons.
The formula to calculate formal charge of the atom is as follows:
Some molecules and ions do not have one unique Lewis structure. The Lewis structures that differ only in the placement of multiple bonds are called resonance structures.
Resonance structures are defined as a set of two or more Lewis structures that collectively describe the electronic bonding. The actual bonding is an average of the bonding in the resonance structures. Also, not all resonance structures contribute equally in every case. Resonance structures that have high formal charges or that place charges of the same sign on adjacent atoms do not contribute to the bonding.
(a)
Answer to Problem 9.69QE
The possible resonance structures are,
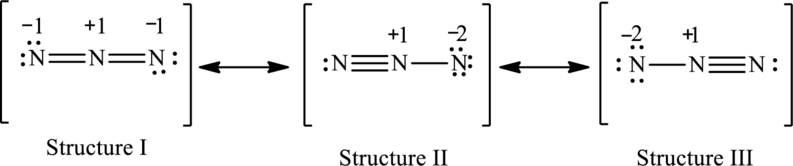
Structure I is more important.
Explanation of Solution
The given skeleton structure is,
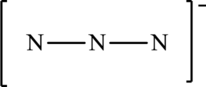
The resonance structures are as follows:
For structure I:
Substitute 5 for valence electrons, 4 for the number of lone pairs of electrons and 4 for the number of shared electrons in equation (1) to calculate the formal charge on first
Substitute 5 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on second nitrogen atom.
Substitute 5 for valence electrons, 4 for number of lone pairs of electrons and 4 for the number of shared electrons in equation (1) to calculate the formal charge on third nitrogen atom.
For structure II:
Substitute 5 for valence electrons, 2 for the number of lone pairs of electrons and 6 for the number of shared electrons in equation (1) to calculate the formal charge on first
Substitute 5 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on second nitrogen atom.
Substitute 5 for valence electrons, 6 for number of lone pairs of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on third nitrogen atom.
For structure III:
Substitute 5 for valence electrons, 6 for the number of lone pair of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on first
Substitute 5 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on second nitrogen atom.
Substitute 5 for valence electrons, 2 for number of lone pairs of electrons and 6for the number of shared electrons in equation (1) to calculate the formal charge on third nitrogen atom.
The possible resonance structures with the formal charges are as follows:
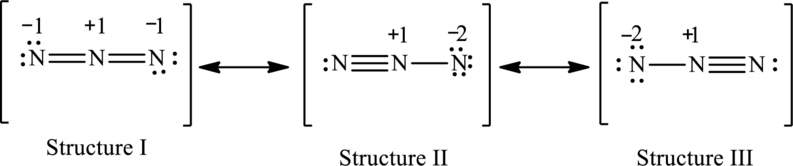
In these resonance structures, structure I have smallest formal charge because Lewis structures that have smallest formal charges are favored.
Hence, structure I is more important.
(b)
Interpretation:
The possible resonance structures for the following structure have to be determined. Also, the resonance structure that is the most important has to be determined.
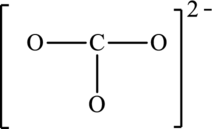
Concept introduction:
Refer to part (a)
(b)
Answer to Problem 9.69QE
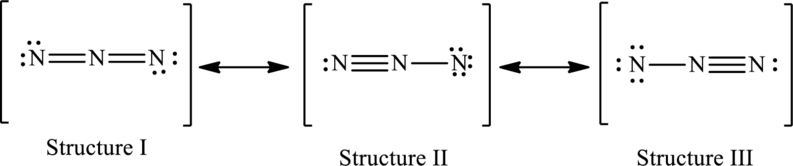
The possible resonance structures are as follows:
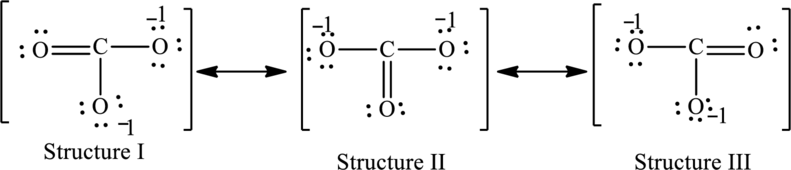
All structures are equally important.
Explanation of Solution
The given skeleton structure is,
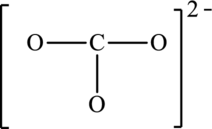
The resonance structures are as follows:
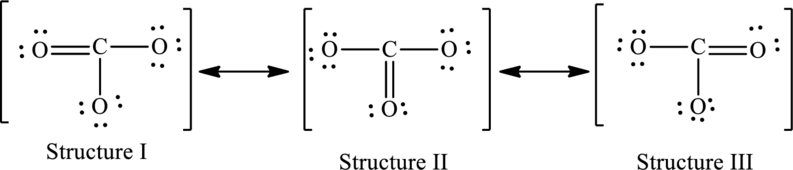
For structure I:
Substitute 4 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on carbon atom.
Substitute 6 for valence electrons, 4 for number of lone pairs of electrons and 4for the number of shared electrons in equation (1) to calculate the formal charge on first oxygen atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on second oxygen atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on third oxygen atom.
For structure II:
Substitute 4 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on carbon atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2for the number of shared electrons in equation (1) to calculate the formal charge on first oxygen atom.
Substitute 6 for valence electrons, 4 for number of lone pairs of electrons and 4for the number of shared electrons in equation (1) to calculate the formal charge on second oxygen atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2for the number of shared electrons in equation (1) to calculate the formal charge on third oxygen atom.
For structure III:
Substitute 4 for valence electrons, 0 for number of lone pairs of electrons and 8 for the number of shared electrons in equation (1) to calculate the formal charge on carbon atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on first oxygen atom.
Substitute 6 for valence electrons, 6 for number of lone pairs of electrons and 2 for the number of shared electrons in equation (1) to calculate the formal charge on second oxygen atom.
Substitute 6 for valence electrons, 4 for number of lone pairs of electrons and 4 for the number of shared electrons in equation (1) to calculate the formal charge on third oxygen atom.
Possible resonance structures are as follows:
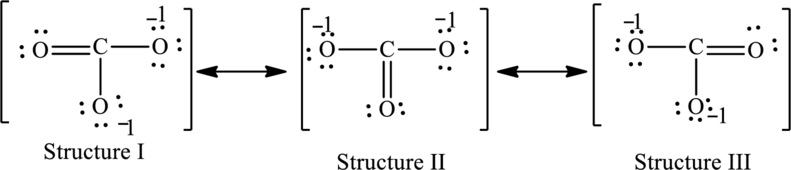
In these resonance structures, all the structures have the same formal charge. Hence, all the structures are equally important.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry: Principles and Practice
- Three resonance forms can be drawn for the molecule N2O. Which resonance form is likely to more closely resemble the structure of this molecule? (a) (b) (c)arrow_forwardAspirin is made from salicylic acid, which has this Lewis structure: (a) Which is the longest carbon-carbon bond? (b) Which is the strongest carbon-oxygen bond? (c) Draw resonance structures for this molecule.arrow_forwardNitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forward
- Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forwardDraw resonance formulas of the phosphoric acid molecule, (HO)3PO. Obtain formal charges for the atoms in these resonance formulas. From this result, which resonance formula would you expect to most closely approximate the actual electron distribution?arrow_forwardDraw resonance structures for each of these ions: NSO− and SNO−. (The atoms are bonded in the order given in each case, that is, S is the central atom in NSO−.) Use formal charges to determine which ion is likely to be more stable. Explain why the two ions cannot be considered resonance structures of each other.arrow_forward
- Given the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forwardDraw all resonance structures for methylisocyanate, CH3NCO, a toxic gas used in the manufacturing of pesticides. Which resonance structures are the most important?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





