
Concept explainers
Interpretation:
Whether each structural formula given is correct as per HONC 1234 rule must be explained. The incorrect structures needs to be corrected.
Concept introduction:
HONC 1234 rule states that in most of the molecule H, O, N and C generally makes 1, 2, 3 and 4 bonds respectively.
Answer to Problem 4E
Compound b and d have violated the HONC 1234 rule.
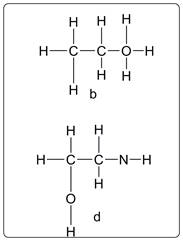
Explanation of Solution
As per the HONC 1234 rule each oxygen (O) atom must form 2 bonds and each nitrogen (N) atom must form three bonds.
As per the structure of b, O has four bonds instead of 2. Thus this is a violation of the rule. Similarly in structure d, N has two bonds instead of three. So it is violation of the rule.
The corrected structures which follow HONC 1234 rule are shown below.
Correct structures are shown as per HONC 1234 rule according to which number of bonds formed by H, O, N and C are 1, 2, 3 and 4 respectively.
Chapter U2 Solutions
Living by Chemistry
Additional Science Textbook Solutions
General Chemistry: Atoms First
Organic Chemistry
Chemistry: Structure and Properties
Chemistry & Chemical Reactivity
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry (9th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





