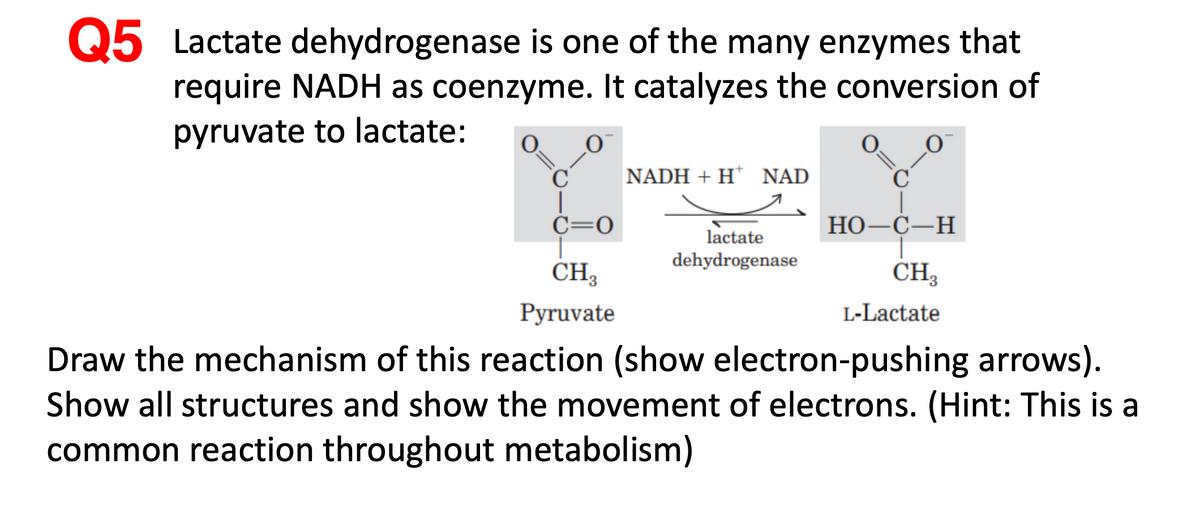
Q5 Lactate dehydrogenase is one of the many enzymes that require NADH as coenzyme. It catalyzes the conversion of pyruvate to lactate: NADH + H* NAD C=0 НО —С—Н lactate dehydrogenase CH3 CH, Рyruvate L-Lactate Draw the mechanism of this reaction (show electron-pushing arrows). Show all structures and show the movement of electrons. (Hint: This is a common reaction throughout metabolism)
Q: Upon digestion of starch, isomaltose (an isomer of maltose), one of its degradation products, is…
A: Isomaltose is the breakdown product of starch. It is an isomer of maltos. It contains two glucose…
Q: dehydrogenase, which converts pyruvate to lactate in the presence of NADH. The velocity of the…
A: Kcat is also known as catalytic constant or turnover number. It can be defined as the amount of…
Q: In beta oxidation of fatty acids in mitochondria, ubiquinone (via FAD / FADH2) from the respiratory…
A: Beta oxidation is the sequential removal of two carbon fragments from the carboxylic end of the…
Q: a) Calculate the enzyme and specific activity of a reaction with 3 pM Hsp90 using the following…
A: An enzyme is a protein that speeds up chemical reactions. Enzymes are involved in all the body's…
Q: Given what you know about the involvement of nicotinamide nucleotides inoxidative and reductive…
A: NADP+ and NADPH are similar to NAD+ and NADH, respectively,except that the NADP+ and NADPH has an…
Q: Consider the malate dehydrogenase reaction from the citric acid cycle. Given the listed…
A: The reaction catalyzed by malate dehydrogenase is given below. Malate + NAD+ ⇌ Oxaloacetate +…
Q: Draw and label a lipid bilayer containing the large mitochondrial trans-membrane protein complexes…
A: Please report to the second half of the question which is the electron flow during photosynthesis…
Q: For the Complex III in the electron transport chain: Complex III step 1: UQH2 is oxidized in a 2…
A: In the question there are two separate processes mentioned Two electron process One Electron…
Q: The glycerol-3-phosphate shuttle can transport cytosolic NADH equivalents into the mitochondrial…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: In living cells, one mole of glucose undergoes a complete oxidation. (a). How many moles of ATP…
A: To get the remaining sub-parts solved, please repost the complete question and mention the…
Q: Q2. How many ATPs are lost in the oxidation of this fatty acid because it is poly- unsaturated? In…
A: First lets calculated total ATP yield if this 18C fatty acid (FA) was completely saturated. 2 ATP…
Q: a) Write the general equation for cellular respiration. i) Based on the equation in 3a, what happen…
A: Introduction Three Processes Are Involved In Cellular Respiration: 1. Glycolysis Is The Process Of…
Q: Oxidative Phosphorylation (Ch. 19) 1. The NADH dehydrogenase complex of the mitochondrial…
A: NADH (nicotinamide adenine dinucleotide hydrogen) dehydrogenase is the first complex in the electron…
Q: Use your knowledge of fat metabolism, glycolysis, the TCA cycle, and oxidative phosphorylation to…
A: Fats are always present in the form of triglycerides or in the esterified form i.e. attached to the…
Q: Describe the process of oxidative phosphorylation. In your description, include the terms NADH,…
A: Glucose is catabolized via glycolysis, pyruvate transformation to acetyl-CoA, TCA cycle, and…
Q: In step 3 of β-oxidation, what reaction is involved? Give the role of NAD+. How many ATPs are…
A: Beta oxidation is the breakdown of fatty acids to produce energy. Most fatty acids are degraded by…
Q: U2, the energy of this reaction is coupled to ne pumping of H+. Drug Y can pick up electrons from…
A: Electron transport chain is the last step of cellular respiration. The first being glycolysis the…
Q: The inhibitor X prevents coenzyme Q (Q) from participating in electron transfer in the electron…
A: The mitochondrion is a double membrane bound structure found in eukaryotic cells that is involved in…
Q: All the dehydrogenases of glycolysis and the citric acid cycle use NAD+ (?′°E′° for NAD+/NADH is…
A:
Q: During electron transfer through the mitochondrial respiratory chain, the overall reaction is NADH +…
A: “The electron transport chain” is found in the inner mitochondrial membrane in eukaryotes. The…
Q: Please compute the standard transformed free energy change as electrons enter the electron transport…
A: Introduction: In the electron transfer chain, NADH donates electrons that enter the respiratory…
Q: Use your knowledge of fat metabolism. glycolysis, the TCA cycle, and axidative phosphorylation to…
A: The cell requires energy from the fats, which are degraded into fatty acid and glycerol. These…
Q: Given what you know about the involvement of nicotinamide nucleotides in oxidative and reductive…
A: The nicotinamide nucleotides are involved in respiration as the electron donor. It is not only the…
Q: . Using electron flow arrows, show electron transfer from nadh to fmn in complex 1 of the electron…
A: The electron transport (ETC) chain is coupled with ATP synthesis. ETC occurs in Cristea…
Q: Calculate the standard free-energy change, deltaG°, for the reaction in which acetaldehyde is…
A: Standard free energy of biochemical reaction is related to standard reduction potential ∆Go = -nFEo…
Q: dtermoine numberof ATPS GENERATED FROM COMPLETE oxidation of fructose-6-phosphate isocitrate…
A: Cellular respiration is a catabolic pathway of the process of metabolism, where a series of chemical…
Q: Pyruvate oxidation yields carbon dioxide and water and liberates energy at the rate of 1142.2…
A: Pyruvate enters TCA cycle and gets oxidized to CO2 and water. Given, Energy released in chemical…
Q: The barbituate drug amytal inhibits complex I from passing electrons to Coenzyme Q. Circle your…
A: According to the question, the barbiturate drug Amytal inhibits the complex I from passing electrons…
Q: Most biochemists agree that the most accurate number of H+ needed to generate one ASP is blank.…
A: Two ATP were formed as a result of glycolysis, two NAD+ were reduced to two NADH + H+, and glucose…
Q: Nicotinamide nucleotide transhydrogenase (NNT) in the inner mitochondrialmembrane catalyzes the…
A: The nicotinamide nucleotide transhydrogenase (NNT) is an enzyme which catalyzes the redox reaction…
Q: Calculate the standard free-energy change, deltaG'o, for the reaction in which acetaldehyde is…
A: Relevant reaction and their standard reduction potential: Acetaldehyde + 2H+ + 2e- →ethanol…
Q: Calculate the standard free-energy change, deltaG'o, for the reaction in which acetaldehyde is…
A: NADH is used as the biological electron carrier and is used for the reduction of Acetaldehyde in…
Q: b) NADH dehydrogenase Q → Cytochrome aa3 → Sequence of complexes in electron transport chain is- a)…
A: Electron transport chain is required for production of ATP from NADH and FADH2, intermediates of…
Q: If a C18 compound is metabolized and enters the citric acid cycle as nine C2 fragments (acetyl…
A: Metabolism is the process of anabolism and catabolism combined. It includes both the generative and…
Q: a With which of the possible fates of pyruvate – acetyl CoA, lactate, and ethanol – is each of the…
A: Note : Hi ! Thank you for the question. We are authorized to answer three subparts at a time. Since…
Q: Given the following information on reduction potentials, calculate the standard free energy in…
A: Gibbs's free energy calculation helps to calculate chemical energy when a bond break or form in a…
Q: 55. Which of the following statements about the glucose-6-P dehydrogenase is INCORRECT? A. It…
A: Enzymes are biocatalysts, which are classified into six classes based on the type of reaction they…
Q: r a 12-carbon saturated fatty acid, calculate the following: er of acetyl COA er of NADH produced…
A: In one pass through the fatty acid oxidation sequence one molecule of acetyl coenzyme A, 2 pairs of…
Q: the complete b-oxidation of stearic acid (18:0) What is the total ATP produced in the citric acid…
A: Stearic acid is a long fatty acid with 18 carbon and its breakdown will produce 9 acetyls CoA’s…
Q: Describe, in simple diagrams and a few words, the chemiosmotic theory for coupling oxidation to…
A: The chemiosmotic theory was first proposed in 1961 that talks about the synthesis of ATP in a…
Q: Explain why the citric acid cycle is considered to be the center of cellular metabolism. at are the…
A: The steps of citric acid cycle were given by Hand kreb in 1931. Acetyl coenzyme A produced in…
Q: Number these compounds in order from lowest to highest actual physiological reduction potential.…
A: Electron transport chain of mitochondria consists of four complexes termed as I, II, III and IV. The…
Q: In the complete b-oxidation of stearic acid (18:0) How many moles of FADH2 are produced? How many…
A: Respiratory chain is a series of complexes that transfer electrons from electron donors to electron…
Q: After pyruvate is formed, it must either be converted to another molecule to enter the citric acid…
A: Glycolysis is a metabolic pathway that converts glucose into pyruvate. After that pyruvate is…
Q: Provide a simplified schematic presentation (metabolic map) of mitochondrial beta-oxidation. Include…
A: Mitochondrial beta oxidation is an aerobic process. oxidation of acyl CoA derivatives catalysed by…
Q: a) Assuming that ubiquinone is unavailable inside of the cell, calculate the AG and the Keq if…
A: 2nd reaction should be reversed for complex I as it in complex I NADH gets converted to NAD+ and…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 4 images

- Q29. FADH2 is similar to NADH in that it is also oxidized by the respiratory electron transport chain. However, oxidation of FADH2 does not yield as much ATP as the oxidation of NADH. Why doesn't oxidation of FADH2 generate as much ATP as oxidation of NADH? A. Energy is more spread out in FADH2. B. FADH2 has fewer C-H bonds than NADH. C. FADH2 oxidation occurs at complex II; it therefore bypasses the proton-pumping of Complex I. D. FADH2 oxidation to FAD releases fewer electrons than the oxidation of NADH to NAD+.Q No 2. During Respiration, various oxidation–reduction reactions occur. Could you expand upon this point further?Calculate the biochemical standard cell potential for the oxidation of NADH by molecular oxygen O2 + 2NADH + 2 H+ → 2H2O + 2NAD+
- Calculate how many moles of ATP can be made in standard conditions when 1 mole of protons flow back into the mitochondrial matrix through the ATP synthase. Assume that: i) the matrix pH is 7.8, ii) the cytosolic pH is 7.2, iii)ΔE=0.168 V (inside negative), iv) T=37°C, and v) the standard free energy change for the hydrolysis of 1 mole of ATP is -31.3 kJ/mol.2. Please, determine the answers of these multiple choices, they’re in a,b,c’s. A) The role of oxygen in oxidative phosphorylation (cellular breathing) is: -To be the last electron acceptor in the electron transfer chain -To hydrolyse carbohydrates - To add hydrogen ions to pyruvic acid at the end of glycolysis -To provide electrons for NADP reduction -To provide hydrogen ions B) Which molecule has the most potential energy? -glucose -phosphate -fructose 1-6 diphosphate -ethanol -I'ATP C) What is the product of fermentation in yeasts? -carbonic acid -oxygen -ATP -lactic acid -ethanol1. Using electron flow arrows, show electron transfer from nadh to fmn in complex 1 of the electron transport chain. 2. From step 3 to 4 of transketolase TPP is eliminated, and S7P is yielded and regenerated. Show steps 3 to 4 with the complete arrow pushing mechanism for both steps and all figures drawn.
- The Relative Efficiency of ATP Synthesis in Noncyclic versus Cyclic Photophosphorylation If noncyclic photosynthetic electron transport leads to the translocation of 7 H+/2e- and cyclic photosynthetic electron transport leads to the translocation of 2 H+/e-, what is the relative photosynthetic efficiency of ATP synthesis (expressed as the number of photons absorbed per ATP synthesized) for noncyclic versus cyclic photophosphorylation? (Assume that the CF1CF0-ATP synthase yields 3 ATP/14 H+.)21. WHICH OF THE FOLLOWING ARE PRODUCTS OF THE CITRIC ACID CYCLE?A. ACETYL COA AND NADHB. CO2 AND FADH2C. ACETYL COA AND CO2D. CO2 AND H20 22. WHICH ARE THE FIRST TWO INTERMEDIATES RESPECTIVELY, IN THE CITRIC ACID CYCLE? A. ISOCITRATE AND SUCCINATEB. ISOCITRATE AND ALPHA-KETOGLUTRATEC. CITRATE AND ISOCITRATED. CITRATE AND ALPHA-KETOGLUTRATE 23. IT IS THE SUM OF ALL THE BIOCHEMICAL REACTIONS THAT TAKES PLACE IN A LIVING ORGANISM?A. ANABOLISMB. METABOLISMC. CATABOLISMD. METABOLIC PATHWAY 24. METABOLIC REACTION IN WHICH LARGE BIOCHEMICAL MOLECULES ARE BROKEN DOWN TO SMALLER ONES?A. CATABOLISMB. METABOLIC PATHWAYC. ANABOLISMD. METABOLISM 25. A METABOLIC REACTION IN WHICH SMALL BIOCHEMICAL MOLECULES ARE JOINED TOGETHER TO FORM LARGER ONES?A. METABOLISMB. CATABOLISMC. METABOLIC PATHWAYD. ANABOLISMBIOC 384 Mitochondrial Electron Transport Q11.3: What explains the observation that FADH2 oxidation yields one less ATP than NADH oxidation by the Electron Transport System?
- ce BiochemistryQ&A Library1. Under aerobic catabolism of glucose, in which compartment of the eukaryotic cell does the following reactions occur? a. Conversion of pyruvate to acetyl CoA. b. Conversion of succinyl-CoA to succinate c. Conversion of NADH to ATP d. Conversion of phosphoglycerate to phosphoenolpyruvate 1. Under aerobic catabolism of glucose, in which compartment of the eukaryotic cell does the following reactions occur? a. Conversion of pyruvate to acetyl CoA. b. Conversion of succinyl-CoA to succinate c. Conversion of NADH to ATP d. Conversion of phosphoglycerate to phosphoenolpyruvate Question 1. Under aerobic catabolism of glucose, in which compartment of the eukaryotic cell does the following reactions occur? (i) Conversion of pyruvate to acetyl CoA. (ii) Conversion of succinyl-CoA to succinate (iii) Conversion of NADH to ATP (iv) Conversion of phosphoglycerate to phosphoenolpyruvate 2. Describe various mechanisms for regulation of…22.The conversion of pyruvate to acetyl-CoA is catalyzed by enzyme pyruvate dehydrogenase. In this reaction ____. Select one: a.NADP+ is reduced to NADPH b.NAD+ is reduced to NADH c.NADH is oxidized to NAD+ d.NADPH is oxidized to NADP+BIOC 384 Citrate Cycle Regulation and Intermediates Q11.1: Explain why a mutation in pyruvate carboxylase that blocks its ability to be regulated by acetyl-CoA results in lower rates of energy conversion via the Citrate Cycle. Hint: see figure 10.36.

