
Genetics: From Genes to Genomes
6th Edition
ISBN: 9781259700903
Author: Leland Hartwell Dr., Michael L. Goldberg Professor Dr., Janice Fischer, Leroy Hood Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 15P
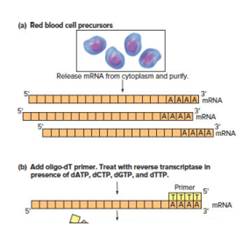
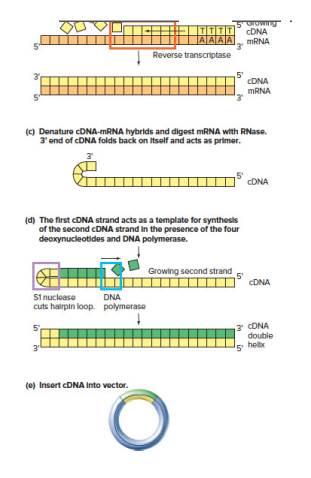
In Problem 14, cDNAs F and G could not be found in cDNA libraries (from any tissue) prepared using the method shown in Fig. 10.4. The reason is that the corresponding transcripts do not have poly-A tails.
| a. | Why is the lack of poly-A tails not surprising in light of your answer to part (d) of Problem 14? |
| b. | Why does the lack of poly-A tails present a difficulty for the method diagrammed in Fig. 10.4? |
| c. | Outline how you might adjust the protocol in Fig. 10.4 so as to find the cDNAs F and G annotated in the Genome Browser |
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The DNA-binding domain of each CREB protein subunit recognizes the sequence 5′–TGACGTCA–3′. Due to random chance, how often would you expect this sequence to occur in the human genome, which contains approximately 3 billion base pairs? Actually, only a few doze genes are activated by the CREB protein. Does the value of a few dozen agree with the number of random occurrences expected in the human genome? If the number of random occurrences of the sequence in the human genome is much higher than a few dozen, provide at least one explanation why the CREB protein is not activating more than a few dozen gene Actually, only a few doze genes are activated by the CREB protein. Does the value of a few dozen agree with the number of random occurrences expected in the human genome? If the number of random occurrences of the sequence in the human genome is much higher than a few dozen, provide at least one explanation why the CREB protein is not activating more than a few dozen gene
As part of a project investigating potential new drug targets in the fight against malaria, you are seeking to clone the gene for a protein from the malaria parasite Plasmodium falciparum. You wish to express this protein in BL21 (DE3) cells, a standard laboratory strain of Escherichia coli. After purification of your protein, you run an SDS-PAGE gel and notice that the major band has lower molecular weight than expected, so you fear you are getting a truncated version.
1. What technique could you use to confirm that you are obtaining a shortened version of your intended protein? explain
As part of a project investigating potential new drug targets in the fight against malaria, you are seeking to clone the gene for a protein from the malaria parasite Plasmodium falciparum. You wish to express this protein in BL21 (DE3) cells, a standard laboratory strain of Escherichia coli. After purification of your protein, you run an SDS-PAGE gel and notice that the major band has lower molecular weight than expected, so you fear you are getting a truncated version.
(a) Give TWO possible causes of your protein becoming truncated. explain
Chapter 10 Solutions
Genetics: From Genes to Genomes
Ch. 10 - Prob. 1PCh. 10 - List three independent techniques you could use to...Ch. 10 - Figure 10.2a has numbers indicating the...Ch. 10 - Which of the enzymes from the following list would...Ch. 10 - Prob. 5PCh. 10 - a. What sequence information about a gene is...Ch. 10 - Why do geneticists studying eukaryotic organisms...Ch. 10 - Consider three different kinds of human libraries:...Ch. 10 - The human genome has been sequenced, but we still...Ch. 10 - This problem investigates issues encountered in...
Ch. 10 - For the sake of simplicity, Fig. 10.4 omitted one...Ch. 10 - Give two different reasons for the much higher...Ch. 10 - Using a cDNA library, you isolated two different...Ch. 10 - The figure that follows shows part of a modified...Ch. 10 - In Problem 14, cDNAs F and G could not be found in...Ch. 10 - Fig. 10.10 presents a model for exon shuffling in...Ch. 10 - An interesting phenomenon found in vertebrate DNA...Ch. 10 - a. If you found a zinc-finger domain which...Ch. 10 - Prob. 19PCh. 10 - In the human immune system, so-called B cells can...Ch. 10 - Chimpanzees have a set of hemoglobin genes very...Ch. 10 - Complete genome sequences indicate that the human...Ch. 10 - On your computers browser, view the page accessed...Ch. 10 - Prob. 24PCh. 10 - Prob. 25PCh. 10 - Certain individuals with mild forms of...Ch. 10 - The 1 and 2 genes in humans are identical in their...Ch. 10 - Prob. 28P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- This problem investigates issues encountered in sequencing the inserts in cDNA libraries.a. If you sequenced many clones individually, wouldn’tyou spend many of your resources inefficiently sequencing cDNAs for the same type of mRNA molecule over and over again? Explain. Does thisapparently inefficient process provide any useful information beyond the sequences of individual mRNAs?b. Suppose that you identified a clone with a cDNA insert that was 4 kb long. You could determine the entire sequence of the clone by shearing the DNA intosmall random fragments, cloning these fragments intoa vector to make a mini-shotgun library, and then sequencing hundreds of these clones to allow the computer to assemble the full sequence of the 4 kb–longinsert. However, this procedure would be inefficient.An alternative that requires many fewer sequencing reactions is called primer walking. This techniqueinvolves the synthesis of additional oligonucleotideprimers corresponding to cDNA sequences you…arrow_forwardGiven the partial transposons DNA sequence 5’-ACCGTATTCGGT-3’ upstream from the central region, assuming both terminal inverted repeats and flanking direct repeats have 6 base pairs, hypothetically write the transposon structure downstream from the central region.arrow_forwardThe following figure shows a screen shot from the UCSC Genome Browser, focusing on a region of the human genome encoding a gene called MFAP3L. (Note hg38 refers to version 38 of the human genome RefSeq)a. Describe in approximate terms the genomic location of MFAP3L.b. Is the gene transcribed in the direction from the centromere-to-telomere or from the telomere-to-centromere?c. How many alternative splice forms of MFAP3L mRNA are indicated by the data?d. How many different promoters for MFAP3L are suggested by the data?arrow_forward
- The following figure shows a screen shot from the UCSC Genome Browser, focusing on a region of the human genome encoding a gene called MFAP3L. (Note hg38 refers to version 38 of the human genome RefSeq)a. Describe in approximate terms the genomic location of MFAP3L.b. Is the gene transcribed in the direction from the centromere-to-telomere or from the telomere-to-centromere?c. How many alternative splice forms of MFAP3L mRNA are indicated by the data?d. How many different promoters for MFAP3L are suggested by the data? (please do not copy and paste the answer from below. i don't think it is correct. a. MFAP3L is mostly found in the nucleus in the genome. It is found on chromosome 4 reverse strand. The protein produced by the gene is found in the cell membrane, and it is positioned on the membrane with the carboxyl side of the protein facing the cytosol. b. The MFAP3L gene is transcribed from the telomere to the centromere. c. According to the data, there are 11 different splice forms…arrow_forwardThe following figure shows a screen shot from the UCSC Genome Browser, focusing on a region of the human genome encoding a gene called MFAP3L. (Note hg38 refers to version 38 of the human genome RefSeq) a. Describe in approximate terms the genomic location of MFAP3L. b. Is the gene transcribed in the direction from the centromere-to-telomere or from the telomere-to-centromere? c. How many alternative splice forms of MFAP3L mRNA are indicated by the data? d. How many different promoters for MFAP3L are suggested by the data?arrow_forwardThe goal of the Knockout Mouse Project is to generate a set of ES cell lines, each with a knockout mutation in a single gene, that collectively contains amutation in every gene in the mouse genome.a. Do you think that it will be possible, for everygene, to generate a heterozygous knockout ES cellline? Explain.b. Do you think that for every heterozygous knockoutES cell line, it will be possible to generate a heterozygous knockout mouse? Explain.c. In fact, investigators failed in their attempts to generate ES cell knockouts for the Fam gene describedin Problem 5. How could these researchers use EScell technology to determine whether Fam is required for eye development in the mouse? Diagrama construct that the researchers could introduceinto ES cells to explore this issue.d. Describe various outcomes that might be obtainedfrom the experiment in part (c) and what you couldconclude in each case.arrow_forward
- The E. coli genome contains approximately 4639 kb. (a) How many copies of the 6-bp recognition sequence for the trp repressor would be expected to occur in the E. coli chromosome? (b) Explain why it is advantageous for the trp repressor to be a dimer that recognizes two adjacent 6-bp sequences.arrow_forwardWhat would be the consequence of inserting 10 random base pairs immediately downstream of the -10 box in a strong promoter? And What sort of lab techniques/experiments could be used to determine these consequences?arrow_forwardThe protein encoded by the cystic fibrosis gene is 1480amino acids long, yet the gene spans 250 kb. How is thisdifference possible?arrow_forward
- in the human gene for the beta chain of hemoglobin, the first 30 nucleotides in the amino acid coding region is represented by the sequence 3'TACCACGTGGACTGAGGACTCCTCTTCAGA-5'. What is the sequence of the partner strand? If the DNA duplex for the beta chain of hemoglobin above were transcribed from left to right, deduce the base sequence of the RNA in this coding region.arrow_forwardLysine 4 of histone H3 (H3K4) is methylated in thenucleosomes of many transcriptionally active genes.Suppose you want to determine all the places in thehuman genome where nucleosomes contain methylated H3K4.a. Starting with an antibody that specifically bindsonly to the tails of histone H3s that have K4 methylation, what kind of experiment would you perform? Outline the major steps of this experiment.b. Do you think that you would get the same results ifyour starting material was skin cells in one experiment and blood precursor cells in a second experiment? Explain.c. Describe a follow-up experiment that could determine if your data from part (a) are consistent withthe idea that H3K4 methylation marks appear onlyat transcriptionally active genesarrow_forwardComparing the -10 regions of two E. coli promoters which have identical -35 regions revealed the sequence TATAAT for the first and GATACT for the second one. Why does the first promoter cause a higher transcription rate than the second one? a. The transcription rate from the first promoter will be higher, because RNA polymerase will bind TATAAT with a higher affinity than GATACT. b. It will be higher, because formation of the open promoter complex is more easily achieved with TATAAT than with GATACT. c. It will be higher, because TATAAT of the -10 region is transcribed into UAUAAU, which forms fewer hydrogen bonds with the template strand than GAUACU. d. a and b, but not c e. a, b, and carrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
An Introduction to the Human Genome | HMX Genetics; Author: Harvard University;https://www.youtube.com/watch?v=jEJp7B6u_dY;License: Standard Youtube License