
Concept explainers
(a)
Interpretation:
The condensed formula of the product of the reaction of ethylene glycol with stearic acid has to be drawn.
Concept Introduction:
Preparation of Esters:
Esters are prepared from the reaction of carboxylic acid and an alcohol, with a loss of water molecule. Esterification is a condensation reaction because a molecule of water is removed during the reaction.
The general preparation of esters is shown below,
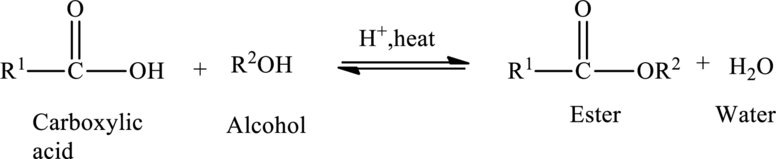
Condensed formula: Condensed formula shows the arrangement of atoms in grouped form.
(b)
Interpretation:
The condensed formula of the product of the esterification reaction of oxalic acid with ethanol has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The condensed formula of the product of the esterification reaction of 1-butanol with propanoic acid has to be drawn.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- The ester with the formula C8H16O2 gives an alcohol and an acid when hydrolyzed. When the alcohol is isolated and oxidized, it forms a ketone. Which of these formulas cannot be the ester?arrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardWrite a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forward
- Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forwarddraw the structure of C8H10NO7P2- and give the general name of the functional grouparrow_forwardWrite a condensed structural formula for a cyclic ketone with the formula C5H8Oarrow_forward
- Write the structural formula of 2-ethyl-1-butenearrow_forwardDraw the condensed structure of the compounds formedby condensation reactions between (a) benzoic acid andethanol, (b) ethanoic acid and methylamine, (c) acetic acidand phenol. Name the compound in each case.arrow_forwardwhich functional groups are in C6H12O2 with it having 1 degree of saturation, and is the molecule hexanoic acid?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





