
Concept explainers
(a)
Interpretation:
The systematic name of the compound
Concept Introduction:
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-ene” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-yne” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Geometrical isomers: In geometrical isomers, atoms have different arrangements on either side of a double bond above or below the ring of a cycloalkane or cycloalkane. If the atoms are present on the same side of the double bond, then it is cis-isomer and if they are present on the opposite side of the double bond, then it is trans-isomer.
(a)
Answer to Problem 11.7E
The systematic name of the compound
The geometrical isomers are not possible in
Explanation of Solution
The given compound is,
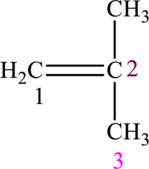
The compound is identified as alkene. The parent chain of the compound is propane. One methyl group is present in the carbon second position and a double bond is seen at the carbon first position. Hence, the systematic name of the compound is
The geometrical isomers are not possible in
(b)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 11.7E
The systematic name of the compound
Cis-
Explanation of Solution
The given compound is,
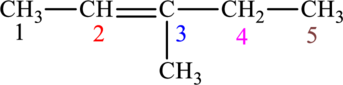
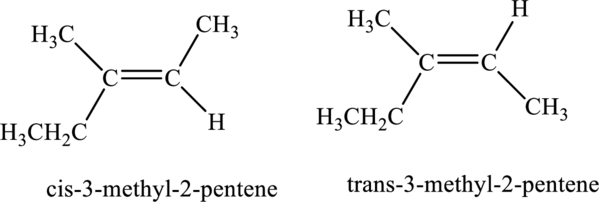
The compound is identified as alkene. The parent chain of the compound is pentane. One methyl group is present in the carbon third position and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
Cis-
(c)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 11.7E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
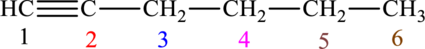
The compound is identified as alkyne. The parent chain of the compound is hexane. One triple bond is present in carbon first position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
(d)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 11.7E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
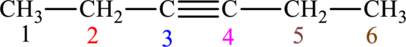
The compound is identified as alkyne. The parent chain of the compound is hexane. One triple bond is present in carbon third position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
(e)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(e)
Answer to Problem 11.7E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
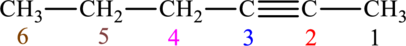
The compound is identified as alkyne. The parent chain of the compound is hexane. One triple bond is present in carbon second position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- Draw and name the five cycloalkane structures of formula C5H10. Can any of these structures give rise to geometric (cis-trans) isomerism? If so, show the cis and trans stereoisomersarrow_forwardHow many straight-chain isomers are there of hexene, C6H12?arrow_forwardwrite the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forward
- An isomer of CH3CH=CHCH2-OH isarrow_forwardwhat are the functional groups in the molecule C3H8O?arrow_forwardComplete and balance the following combustion reactions. Assume that each hydrocarbon is converted completely to carbon dioxide and water. (a) Propane + O2 (b) Octane + O2 (c) Cyclohexane + O2 (d) 2-Methylpentane + O2arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





