
Concept explainers
(a)
Interpretation:
The product formed from the condensation reaction of butanoic acid and 2-propanol has to be drawn.
Concept Introduction:
Ester:
The structure of an ester is,
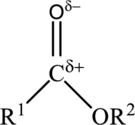
The carbonyl group of the ester is polar and could participate in dipole-dipole attractions. Hydrocarbon chains flank the carbonyl group
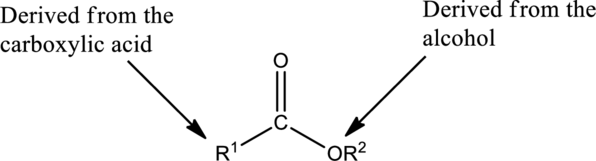
Preparation of Esters:
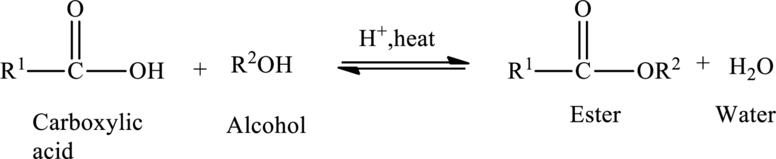
Esters are prepared from the reaction of carboxylic acid and an alcohol, with a loss of water molecule. Esterification is a condensation reaction because a molecule of water is removed during the reaction.
The general preparation of esters is shown below,
(b)
Interpretation:
The product formed from the condensation reaction of ethanoic acid and 1-pentanol has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product formed from the condensation reaction of hexanoic acid and methylethyl
Concept Introduction:
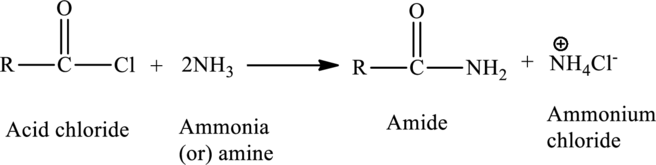
Amides can be prepared in a reaction between a carboxylic acid and an amine. The acid derivative can be either an acid chloride or an acid anhydride.
The general reaction is,
(d)
Interpretation:
The product formed from the condensation reaction of ethanoic acid and propylamine has to be drawn.
Concept Introduction:
Refer to part (d).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forwardDraw the condensed structure of the compounds formedby condensation reactions between (a) benzoic acid andethanol, (b) ethanoic acid and methylamine, (c) acetic acidand phenol. Name the compound in each case.arrow_forwardGlucose, C6H12O6, contains an aldehyde group but exist predominantly in the form of the cyclic hemiacetal show below. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. Identify which carbon provides the —OH group and which provides the —CHO? Give a functional isomer of glucose and draw its structure.arrow_forward
- Write structures and describe thephysical properties of carboxylic acidsarrow_forwardWrite the structure of the following compounds: a. cyclohexyl propanoate, b. 4-methylheptanonitrile, c. butanoic benzoic anhydride, d. N,N-dibenzylmethamide, e. 3-methylhexanoyl chloride.arrow_forwardWrite the structural formula of methylcyclopentenearrow_forward
- How to prepare ethane bromine through: a) a substitution reaction b) an addition reaction c) How many grams add 1 liter of ethene, but 1 liter of C2H2?arrow_forwardIdentify whether oxidation or reduction is needed to interconvert alkanes, alcohols,aldehydes, ketones, and acids, and identify reagents that will accomplish the conversionarrow_forwardGive a chemical test to distinguish between 2-Pentanol and 3-Pentanol.arrow_forward
- If 3-Mehtylhexanoic acid reacts with 3-pentanol, draw the resulting skeletal structure and provide its name?arrow_forwardwrite the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forwardWhat function does sulfuric acid have in the reaction with 1-butanol to form 1-bromobutane?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

