
Concept explainers
(a)
Interpretation:
The reaction and the alcohol that is used for the preparation of ethanal have to be given.
Concept Introduction:
The oxidation of a primary alcohol produces an aldehyde.
The generalized equation is written as,
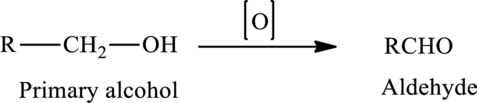
The oxidation of a secondary alcohol produces a ketone.
The generalized equation is written as,
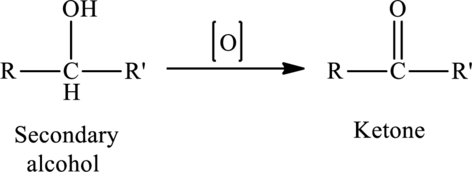
(b)
Interpretation:
The reaction and the alcohol that is used for the preparation of 2-octanone have to be given.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The reaction and the alcohol that is used for the preparation of 5-methyloctanal have to be given.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- 5. How many isomers are there Of C6H14? 6. Aside from a halogenated alkane, what other product results from the bromination of hexane? Write the formulaarrow_forwardPropene undergoes a hydration reaction with water in the presence of an acid. a. There are two possible products for this reaction, both with the formula C3H8O. Show their structural formulas and names. b. Which of the two products do you predict will form. Explainarrow_forwardThe ester with the formula C8H16O2 gives an alcohol and an acid when hydrolyzed. When the alcohol is isolated and oxidized, it forms a ketone. Which of these formulas cannot be the ester?arrow_forward
- Glucose, C6H12O6, contains an aldehyde group but exist predominantly in the form of the cyclic hemiacetal show below. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. Identify which carbon provides the —OH group and which provides the —CHO? Give a functional isomer of glucose and draw its structure.arrow_forwardwrite the structure formulas of alkanes with molecular formula C6H14, which with chlorine give: a) three monochlorinated isomers? b) five monochlorinated isomers c) only two monochlorinated isomersarrow_forwardEnter the molecular formula for butane, C4H10?arrow_forward
- Suggest a method of preparing ethyl benzene, starting with benzene and ethylene as the only organic reagents.arrow_forwardWhich of the following structural features could be found in a compound with formula C7H12?arrow_forwardFrom the spectral data that was given, identify the structure of the ester product. The mass is 116 amu and the molecular formula is C6H12O2. Based on your answer what is the structure of the starting alcohol?arrow_forward
- Which alcohols can be prepared as a single product by hydroboration– oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?arrow_forwardWhich alcohols can be prepared as a single product by hydroboration–oxidation of an alkene? Which alcohols can be prepared as a singleproduct by the acid-catalyzed addition of H2O to an alkene?arrow_forwardOne mole of an unknown hydrocarbon, compound C, in the presence of a platinum catalyst, adds 98.9 L of hydrogen, measured at 744 mm Hg and 22 degrees C , to form a saturated alkane which contains one ring. When one mole of compound C is reacted with ozone, followed by reduction with (CH3)2S , four moles of only one product was formed, whose condensed molecular formula is CHO -CHO. Give the structure of compound C. Explain your reasoningarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


